
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.SE, Problem 35AP
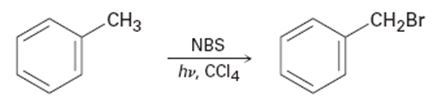
Alkylbenzenes such as toluene (methylbenzene) react with NBS to give products in which bromine substitution has occurred at the position next to the
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The rate law for addition of Br2 to an alkene is first orderin Br2 and first order in the alkene. Does this informationsuggest that the mechanism of addition of Br2 to analkene proceeds in the same manner as for addition of HBr?Explain.
b) Fill in the gaps by drawing the structures of the missing reagents, intermediates and/or
products of the following electrophilic aromatic substitutions. Chemical formulas have been
added for guidance. .
Step 1
Step 2
CH3
HNO.
1.
CH;NO2
C;H3N
Pd/C
NO2
3
1
2
4
CH3
Brz FeBr
C3H;Bro
6
Using resonance structures, justify whether the acetyl group of compound 5 will direct the
bromination reaction to the meta or ortho/para positions
(a) In an acid-catalyzed hydration, one of the following 10 carbon alkynes is expected to produce a single
hydration product? Select the correct alkyne and draw the structure of the hydration product that is formed from
this alkyne.
(I) 2-decyne; (II) 3-decyne; (III) 4-decyne; (IV) 5-decyne; (V) none of these will give a single hydration
product.
(b) The reaction shown below gives Compound X as the major product. The mass spectrum of X is shown.
Br2, H20
Compound X
100 -
MS-IW-5644
80
60
40 -
20 -
20
40
60
80
100
120
140
160
180
200
220
m/z
Considering the reactions of alkynes and the MS data, de duce which of following structures corresponds to X:
Br
Br
HO,
IV
V
I
II
II
Support your answer with a reaction mechanism for fomation of X and identification of relevant peaks in the
mass spectrum.
12
Relative Intensity
Chapter 10 Solutions
Organic Chemistry
Ch. 10.1 - Prob. 1PCh. 10.1 - Draw structures corresponding to the following...Ch. 10.2 - Prob. 3PCh. 10.2 - Taking the relative reactivities of 1°, 2°, and...Ch. 10.4 - Prob. 5PCh. 10.4 - The major product of the reaction of...Ch. 10.4 - Prob. 7PCh. 10.5 - Prob. 8PCh. 10.6 - Prob. 9PCh. 10.6 - How might you replace a halogen substituent by a...
Ch. 10.7 - How would you carry out the following...Ch. 10.8 - Rank both sets of compounds in order of increasing...Ch. 10.8 - Tell whether each of the following reactions is an...Ch. 10.SE - Prob. 14VCCh. 10.SE - Prob. 15VCCh. 10.SE - Prob. 16VCCh. 10.SE - Draw the electron-pushing mechanism for each...Ch. 10.SE - Draw the electron-pushing mechanism for the...Ch. 10.SE - The formation of Br2 from NBS first involves the...Ch. 10.SE - In light of the fact that tertiary alkyl halides...Ch. 10.SE - Alkyl halides can be reduced to alkanes by a...Ch. 10.SE - Name the following alkyl halides:Ch. 10.SE - Prob. 23APCh. 10.SE - Draw and name all of the monochlorination products...Ch. 10.SE - How would you prepare the following compounds,...Ch. 10.SE - Prob. 26APCh. 10.SE - A chemist requires a large amount of...Ch. 10.SE - What product(s) would you expect from the reaction...Ch. 10.SE - What product(s) would you expect from the reaction...Ch. 10.SE - What product would you expect from the reaction of...Ch. 10.SE - Rank the compounds in each of the following series...Ch. 10.SE - Which of the following compounds have the same...Ch. 10.SE - Tell whether each of the following reactions is an...Ch. 10.SE - Prob. 34APCh. 10.SE - Alkylbenzenes such as toluene (methylbenzene)...Ch. 10.SE - Prob. 36APCh. 10.SE - Prob. 37APCh. 10.SE - Prob. 38APCh. 10.SE - Prob. 39APCh. 10.SE - Prob. 40APCh. 10.SE - The syntheses shown here are unlikely to occur as...Ch. 10.SE - Why do you suppose its not possible to prepare a...Ch. 10.SE - Prob. 43APCh. 10.SE - Identify the reagents a–c in the following...Ch. 10.SE - Prob. 45APCh. 10.SE - Prob. 46APCh. 10.SE - Prob. 47APCh. 10.SE - The relative rate of radical bromination is...Ch. 10.SE - Prob. 49APCh. 10.SE - Predict the product and provide the entire...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the following molecules, show how the aryl substituents impact the electron density at the unique carbons around the aromatic ring by showing all unique resonance structures, including appropriate formal charges. OMe Me MeO Mearrow_forward(3) HI does not undergo free radical addition with 1-butene, even in the presence of a peroxide. Please provide an explanation by analyzing the propagation steps if the same radical reaction occurred with HI. Besides the bond dissociation enthalpies in the appendix, the following approximate bond dissociate enthalpies might be useful to you: ~310 kJ/mol The component of C=C CH3CH₂CH₂CH₂-Br CH3CH₂CH₂CH2-I -290 kJ/mol (C-Br bond) ~235 kJ/mol (C-I bond)arrow_forwardInterpret the acidity of alcohols on the basis of ground-state polarization and stability of the alcoholate anion(indicate and give symbols for bond polarization)! Compare the relative acidity of ethanol and 2-fluoroethanol!arrow_forward
- (b) The activating and deactivating groups could affect the position(s) of the next incoming group(s) to the benzene ring. Based on the structure below, analyze and explain the group(s) on the benzene ring is activating or deactivating group. Then, identify the product(s) formed from the following reactions. NH, CC, CH;CH,COCI AICI, (i) NH, HNO, H,SO, (ii) H Br AICI, (iii) Page 3 of 4arrow_forwardb) Fill in the gaps by drawing the structures of the missing reagents, intermediates and/or products of the following electrophilic aromatic substitutions. Chemical formulas have been added for guidance. Step 1 Step 2 CH3 HNO3 H2 1. C6H5NO2 C,HgN H2SO4 `NO2 Pd/C 1 2 4 CH3 Br2/FeBr3 C3H;Bro 5 6 Using resonance structures, justify whether the acetyl group of compound 5 will direct the bromination reaction to the meta or ortho/para positions.arrow_forward(1) Predict the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. How many isomers can be formed in each case? (2) Addition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Draw structures for the two products, and give a mechanistic explanation for their formation.arrow_forward
- 6. (Chapter 15-Q37) Indole is an aromatic heterocyclic that has a benzene ring fused to a pyrrole ring. Answer the following questions. Indole 6(a) What is the hybridization of N in this molecule? = 6(b) How many pi electrons N contributes to the ring? = 6() Which orbitals contribute to form a sigma bond between N and H in this molecule? = 6(c) What is the electronic relationship of Indole to naphthalene? Give the answer by comparing number of rings and number of pi electrons in both compounds, write x rings, y pi electrons=|arrow_forwardCH3 Br,/FeBr3 C3H;BrO 5 Using resonance structures, justify whether the acetyl group of compound 5 will direct the bromination reaction to the meta or ortho/para positions.arrow_forwardDefine the Mechanism of the Radical Addition of HBr to an Alkene ?arrow_forward
- Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions -X티 Hö: H-O -CH3 -CH3 H30*arrow_forwardAcrolein and 1,3-cyclohexadiene react in a one-step concerted manner to yield a single product. Give the structure of the product. What kind of reaction is this an example of? In terms of this reaction, how would you classify acrolein? How would you classify 1,3-cyclohexadiene? Hint: acrolein is not a systematic name so you may need to look up its structure if you are not already familiar with it.arrow_forwardParagraph Styles Edi 7. Answer ALL parts of the question (a) Predict the organic products formed when 3-methoxybenzaldehyde is heated with formaldehyde in the presence of concentrated sodium hydroxide (after final acidification with HCI) (Scheme Q7a): 1. NaOH, heat MeO, H. H. 2. HCI Aromatic (Scheme Q7a) (b) Give the full name of this reaction. (c) (1) Identify the electrophile in the hydride transfer step to form the aromatic product. (ii) Draw a mechanism for the reduction of this electrophile.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY