
Concept explainers
a)
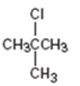
Interpretation:
How to prepare tert-butyl chloride from tert-butylalcohol is to be stated.
Concept introduction:
tert-Alcohols when treated with HCl, HBr or HI in ether at 0oC give the corresponding tert-
To state:
How to prepare tert-butyl chloride from tert-butylalcohol.
b)
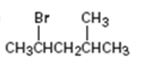
Interpretation:
How to .prepare 2-bromo-4-methylpentane from the corresponding alcohol is to be stated.
Concept introduction:
A secondary alkyl bromide is required. Secondary alkyl halides can be prepared by treating the alcohol required with PBr3 in ether solution.
To state:
How to prepare 2-bromo-4-methylpentane from the corresponding alcohol.
c)
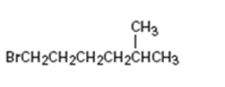
Interpretation:
How to .prepare 1-bromo-5-methylhexane from the corresponding alcohol is to be stated.
Concept introduction:
A primary alkyl bromide is required. Primary alkyl halides can be prepared by treating the alcohol required with PBr3 in ether solution.
To state:
How to prepare1-bromo-5-methylhexane from the corresponding alcohol.
d)
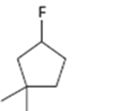
Interpretation:
How to .prepare 1-fluoro-3,3-dimethylcyclopentane from the corresponding alcohol is to be stated.
Concept introduction:
A secondary alkyl fluoride is required. Secondary alkyl fluorides can be prepared by treating the alcohol required with diethylaminosulphur trifluoride and HF in pyridine.
To state:
How to prepare1-fluoro-3,3-dimethylcyclopentane from the corresponding alcohol.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Organic Chemistry
- Starting with cyclohexane, how could the following compounds be prepared?arrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forwardSynthesize the following compounds from the given starting material. You can add on any alkyl/ alkyl halide under 8 carbons or any necessary inorganic reagent needed (this includes triphenyl phosphine (Ph3P). Please draw all intermediates and reagents necessary to get to the productarrow_forward
- Indicate how the following compounds can be synthesized from cyclohexanone and any other necessary reagents:arrow_forwardWhy can’t 1-methylcyclohexanol be prepared from a carbonyl compound by reduction?arrow_forwardwhat product would you expect from reaction of cyclohexane with hbr with hclarrow_forward
- Using bromocyclohexane as a starting material, how could you synthesize the following compounds?arrow_forwardSynthesize the following compounds using cyclohexanol, or any organic reagent that has 4 or fewer carbons, and any common reagents A) но, B) онarrow_forwardWhat reagents are needed to carry out the following syntheses?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





