
Concept explainers
a)
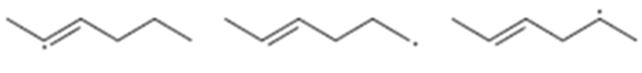
Interpretation:
The radicals given are to be arranged from most stable to least stable.
Concept introduction:
The stability of different radicals is in the order
Benzylic > allylic > tertiary > secondary > primary > methyl.
To sort:
The radicals given from most stable to least stable.
b)
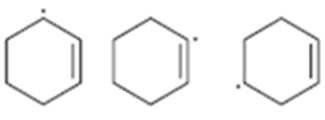
Interpretation:
The radicals given are to be arranged from most stable to least stable.
Concept introduction:
The stability of different radicals is in the order
benzylic > allylic > tertiary > secondary > primary > methyl.
To sort:
The radicals given from most stable to least stable.
c)
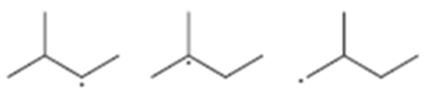
Interpretation:
The radicals given are to be arranged from most stable to least stable.
Concept introduction:
The stability of different radicals is in the order
benzylic > allylic > tertiary > secondary > primary > methyl.
To sort:
The radicals given from most stable to least stable.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Organic Chemistry
- Which of the drawing below is the most stable if the radical carbon is changed to a carbocation? CH3 CH3 O CH₂ CH 0 CH3arrow_forwardMost stable out of each pairarrow_forward3. For the following structures: CH3a C-CH,CH3 C Hy' H (a) Determine whether each radical is a primary, secondary, or tertiary radical. (b) Rank these radicals in terms of their relative stability (1 = most stable to 3 = least stable). 4 Draw the (2) banoggata 1oarrow_forward
- Select the pair of molecules that produce the fastest Diels-Alder reaction and then draw the major organic product(s) formed from them. Add wedges or dashes for the major product formed. CN NH₂ CI Me Me MeO CI Rarrow_forwardNumbering 1-4, rank the carbocations from most stable to least stable.arrow_forwardOrder the following in increasing priority A. -C, -CH, -OL B. -CH3, -CH2OH, -CH2CH3 C. -C≡CH, -CH゠CH2, -CH゠Oarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





