
Concept explainers
a)
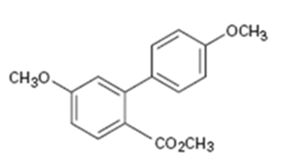
Interpretation:
How the biaryl compound shown can be prepared using Suzuki-Miyaura reaction is to be shown.
Concept introduction:
In Suzuki-Miyaura reaction biaryl compounds are prepared by a coupling reaction between
To show:
How the biaryl compound shown can be prepared using Suzuki-Miyaura reaction.
b)
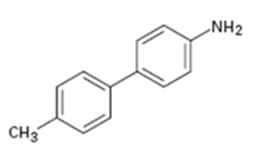
Interpretation:
How the biaryl compound shown can be prepared using Suzuki-Miyaura reaction is to be shown.
Concept introduction:
In Suzuki-Miyaura reaction biaryl compounds are prepared by a coupling reaction between aromatic or vinyl substituted boronic acid with an aromatic or vinyl organo-halide in the presence of Pd catalyst.
To show:
How the biaryl compound shown can be prepared using Suzuki-Miyaura reaction.
c)
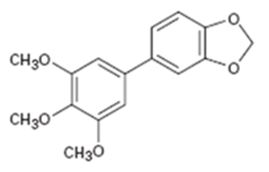
Interpretation:
How the biaryl compound shown can be prepared using Suzuki-Miyaura reaction is to be shown.
Concept introduction:
In Suzuki-Miyaura reaction biaryl compounds are prepared by a coupling reaction between aromatic or vinyl substituted boronic acid with an aromatic or vinyl organo-halide in the presence of Pd catalyst.
To show:
How the biaryl compound shown can be prepared using Suzuki-Miyaura reaction.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Organic Chemistry
- A step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forwardEthylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forwardShow how the following compound could be prepared by a Suzuki reaction (Bn = benzyl).arrow_forward
- How might you convert geraniol into either ethyl geranylacetate or geranylacetone?arrow_forwardUsing your reaction roadmap as a guide, show how to convert butane into butanal. Show all reagents needed and all molecules synthesized along the way.arrow_forwardIdentify the best reagents to complete the following reaction. HO, CIarrow_forward
- how to synthesize 2-phenylclohexanone from cyclohexanone?arrow_forwardAcetic acid has been mixed with isoamyl alcohol to produce isoamyl acetate giving off a banana smell. Propose a reaction mechanism for this reaction.arrow_forwardH₂C ཏཱཏི 1 ནི OH 1. Br2, PBг3 2. H₂O H3C OH Br The a-bromination of carbonyl compounds by Br2 in acetic acid is limited to aldehydes and ketones because acids, esters, and amides don't enolize to a sufficient extent. Carboxylic acids, however, can be a-brominated by first converting the carboxylic acid to an acid bromide by treatment with PBr3. Following enolization of the acid bromide, Br2 reacts in an a-substitution reaction. Hydrolysis of the acid bromide completes the reaction. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions :0: H3C Br Br + :::OH2 Br H₂O H3C Br заarrow_forward
- Starting from cyclohexanone, show how to make the two given products, include conditions to use in the reactions. CH3 CH3arrow_forwardReaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0 °C, followed by treatment with N,N-diethylaniline.arrow_forwardIdentify the best reagents to complete the following reaction. Options are included.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

