
Interpretation: The most likely hydride shift that will occur for each of the below carbocations should be depicted with curved arrow and reason behind lowered potential energy for thus newly formed carbocation should be explained.
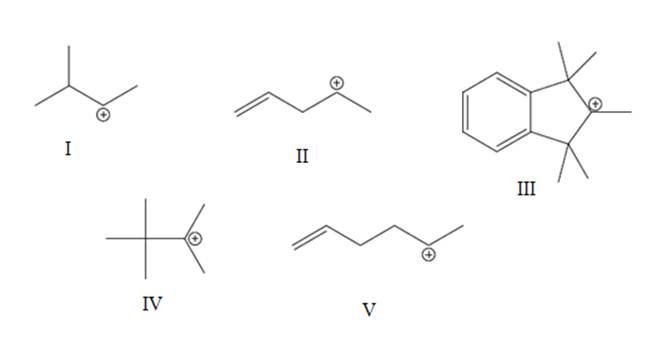
Concept introduction: Carbocation is a general term employed for a postively charged carbon irrespective of valency of carbon. In carbocation, carbon is bonded to 3 atoms or groups and has only sextet of electrons so it behaves as an electron-deficient species. It is
The order of relative stability of various possible carbocation species is as follows:
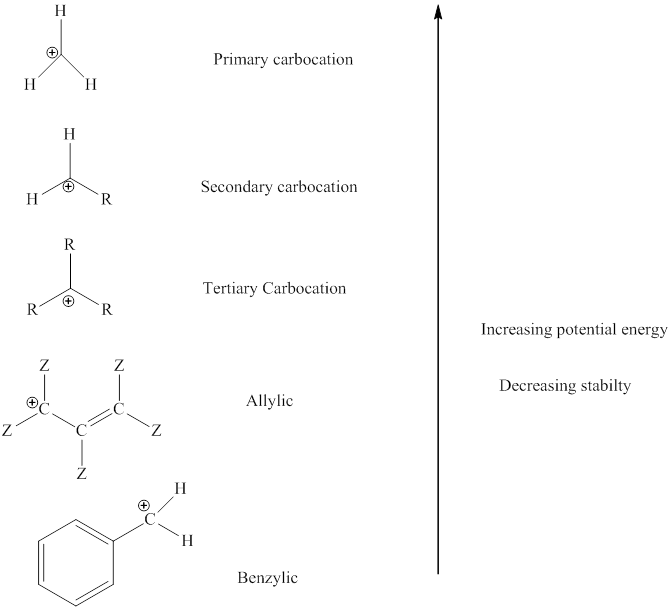
Thus, whenever possibility to attain lower energy by rearrangement is there hydride or alkyl shift may occurs and results in more stable carbocation. This type of rearrangement is highly favorable in polar solvents.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- Draw both resonance structures of the most stable carbocation intermediate in the reaction shown + HCI [Review Topics] [References] Draw a structural formula for the product of this Diels-Alder reaction, including all stereoisomers of the product. a COOEt CHO COOEtarrow_forwardConsider the molecule given below. In theory, there are only two inequivalent hydrogens in this molecule that could be substituted by Br in a free radical bromination – circle them. Put an asterisk to mark the one most likely to be substituted first. However, there are 5 possible products from free radical bromination. Draw all the products and show using arrow formalism how the intermediate radicals leading to these products formedarrow_forwardFor each of the following draw the major product and a mechanism to account for its formation. Be sure to include stereochemistry (if appropriate), arrows to show the movement of electrons and all formal charges.arrow_forward
- why does this carbocation go through rearrangemnt by a hydride shift?arrow_forwardPlease explain the reaction mechanism above to show how product is formed. The product is the borate coplymer .( Please include ALL lone pairs with the structures and provide an explanation of what is happening)arrow_forward9. For the following carbocations: 1) determine if they rearrange, and 2) if they rearrange, draw the expected product and the curved arrows to complete the rearrangement.arrow_forward
- Which of the following carbocations can rearrange to a more stable ion? Propose structures for the likely rearrangement products.arrow_forwardIllustrate the carbocation pathway for the following reactions on the picture below.arrow_forwardCompound A reacts with B in the presence of sodium hydride to give C as the majority product.1. Give the detailed mechanism of the formation of C and draw the product C in the box above.2. Draw the energy diagram of the reaction. Indicate with an arrow the rate-determining step of the reaction then draw its transition state3.The rate of the reaction observed is 5 x 10-2 M.s-1 when the concentrations of A and B are 0.2 M and 0.1 M respectively.Determine the rate of the reaction if the concentrations of A and B are now 0.3 M and 0.2 M respectively. Show your calculationarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

