
Concept explainers
a)
Interpretation: Each transition state in below diagram should be marked with double dagger
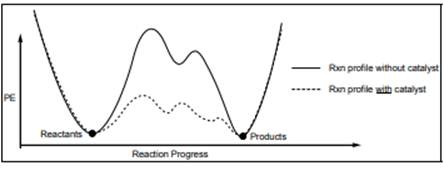
Concept introduction: Energetics of a reaction describes the energy associated with different components during the process of reaction. Energy diagram is a plot of energy along
b)
Interpretation: Number of steps in each reaction below should be determined.
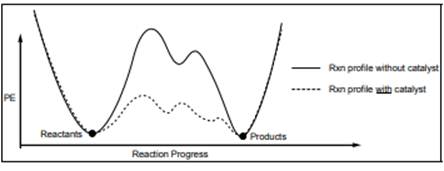
Concept introduction: Energetics of a reaction describes the energy associated with different components during the process of reaction. Energy diagram is a plot of energy along
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- For the reaction can someone please synthesize the given products from the given reactants. Multiple reactions/steps will be needed (the arrows designate the minimum number of steps). Also, for the 1st step (reaction) in each synthesis, can you please draw an energy diagram showing the correct number of hills and valleys for that step’s mechanism?arrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Given the Reaction Energy Diagram, below, identify the following: a. How many steps does the reaction have?b. Which step is the rate determining step?c. Is the overall reaction exothermic or endothermic?d. Is the 2nd step exothermic or endothermicarrow_forward6. a) Based on stability, predict the side of the equilibrium that will be favored. b) Draw a reaction coordinate diagram. H H Loo d Lo NH2 HO NHarrow_forward
- Consider the reaction energy diagram shown below. The reaction shown is reversible, it can proceed from reagents-> products or produCts->reagents. In which direction do you think the reaction has a higher rate constant, k? reactants Reaction pregresk O a. both k's are the same. b. reactants-> products has the higher k. C. reactants to products always has the larger k d. products -> reactants has the higher k. Posential cncrgyarrow_forward3. a) Draw a reaction coordinate diagram of an exothermic reaction, A – B that occurs in a single step. b) In the above reaction, which molecules will have more kinetic energy – A or B? c) If you use a catalyst to speed up the reaction, how will that affect reaction coordinate?arrow_forwardReaction coordinate diagrams: Two-step diagram. Label the reactant (R), product (P), intermediate (I"), and transition states (TS; and TS2), plus the axis with appropriate units/labels. 1) Break the diagram up into 2 steps. Which one is the slow step? The fast step? 2) Endothermic or exothermic? Suppose a is added to step one. Sketch the effect of this addition on the diagram. You can identify the intermediates and by remembering: Intermediates are and then Catalysts are added to a reaction (like a reactant), and are at the end. Practice: Identify the reactants, products, intermediate(s), and catalyst(s). Put a star next to the rate-determining step. Step 1 A+B AB (fastest) Step 2 C + AB → ABC Step 3 ABC + D→ ACD + B (fast) diyarrow_forward
- Use the energy diagram for the reaction A D to answer the questions. How many transition states are there in the reaction? transition states: How many intermediates are there in the reaction? D Reaction progress intermediates: Which step of the reaction is the fastest? Which step of the reaction has the smallest rate constant? Assume the frequency factor (A) is the same for for each elementary reaction. Is the overall reaction exothermic or endothermic? O exothermic O cannot determine from the diagram O endothermic Potential energyarrow_forward5. Draw the energy diagram for the following reaction in the reaction coordinate drawn below. HCI + H20 –→ H3O+ + Cl In your energy diagram, account for the fact that a) the reaction is very fast b) the reaction is strongly exergonic Clearly label all reactants, products, transition states, intermediates, and their energies.arrow_forwardConsider the energy diagram in Figure 6. Select the correct statements. * A- This reaction proceeds through a mechanism with three transition states. B- Step 1 determines the rate of the overall reaction. C- The first reactive intermediate is more stable than the second reactive intermediate. D- This reaction proceeds through a mechanism that has three elementary steps. E- The overall reaction is endothermicarrow_forward
- Consider the following energy diagram. Energy Reaction coordinate a. How many steps are involved in this reaction? b. Label AH and E₂ for each step, and label AH overall- c. Label each transition state. d. Which point on the graph corresponds to a reactive intermediate? e. Which step is rate-determining? f. Is the overall reaction endothermic or exothermic?arrow_forwardRank the following in order of increasing rate in a unimolecular reaction. (1=slowest ... 3=fastest)arrow_forward2. Draw an approximate energy diagram for the following reaction a. It has two transition states and one intermediate b. The overall reaction is exothermic (exergonic) c. The first step is endothermic (endogonic) and fast d. The second step is exothermic (exergonic) and slow.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
