
Concept explainers
Interpretation: The carbocation carbon that is farthest to have an octet should be identified with proper reasons.
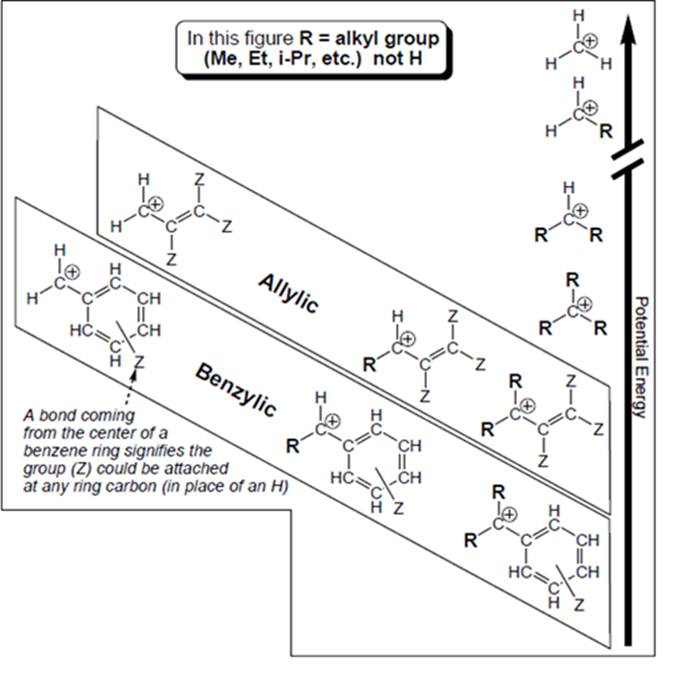
Concept introduction: Organic compounds are covalent in nature that undergoes a reaction by heterolytic cleavage or homolytic cleavage.
In heterolytic cleavage, shared pair of electrons is taken away by one of the atoms which result in charged species. In homolytic cleavage, shared pair of electrons are equally distributed between two atom that results in free radicals.
Carbocation is a general term employed for a postively charged carbon irrespective of valency of carbon. In carbocation, carbon is bonded to 3 atoms or groups and has only sextet of electrons so it behaves as an electron-deficient species. It is
A carbocation is classified as
Explanation of Solution
Allylic and benzyl carbocations are primary carbocation but are stable due to resonance effect. In both cases the vacant
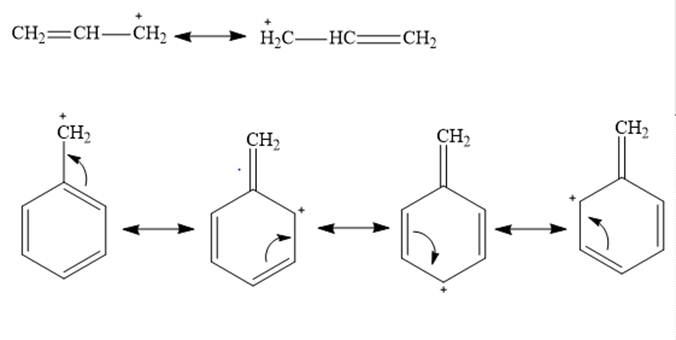
In both benzyl cation and allylic cation, delocalization occurs through resonance effect but allylic cation has 2 contributing structures whereas benzyl cation has 4 contributing structures. Greater the contributing structures greater will be stability. Therefore benzyl cation is more stable than allylic cation.
Outside box methyl carbocation, ordinary primary, secondary and tertiary carbocations are stabilized by electron releasing nature of alkyl groups. This electron releasing nature is due to inductive effect of alkyl groups. They do not exhibit any resonance.
In general, resonance effect dominates over inductive effect. The carbocations that involve delocalization through resonance effect are more stable than those where inductive effect causes stabilization.
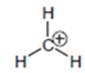
Hence, methyl carbocation is farthest to have an octet as electron releasing nature of hydrogen is less than methyl group so is the inductive effect and it involves no resonance stabilization.
The carbocation that is farthest to have an octet is as follows:
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- Draw thee other resonance structures for the following carbocation. Use electronpushing arrows in order to derive one resonance structure from another. Indicate theformal charge on each atom in your structures.arrow_forwardWhich numbered bond will have the maximum dissociation energy? Which will have the minimum? If reacted with Cl2 which bond would react fastest? Which would react slowest?arrow_forwarddraw the following compound Plz do Asap...!arrow_forward
- B1) Draw all the Resonance structures for Cycloheptatrienyl anion, using arrows show the flow of electrons (delocalization) in each resonance structure.arrow_forwardThe following is generic depiction of a reaction using the curve arrow formalism. -D Which of these statements is not correct for this reaction? Electrons move from C to B. Electrons move from B to A. O In the products, a bond forms between C and B. O In the products, A would have a positive charge.arrow_forwardDraw a resonance structure, complete with all formal charges and lone (unshared) electron pairs, that shows the resonance interaction of the cyano with the ortho position in benzonitrile. CEN benzonitrile • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • In cases where there is more than one answer, just draw one.arrow_forward
- Draw the contributing structure that results from resonance indicated by the curved arrow(s). H- - • You do not have to consider stereochemistry. Explicitly draw all H atoms. • You do not have to include lone pairs in your answer. P opy astearrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps. H HH Select to Add Arrows O..I H3O+ heat HH Select to Add Arrows H3O+ H heat I.O. Please select a drawing or reagent from the question areaarrow_forward4. Circle any n-conjugated portions in the molecules below. Draw all resonance structures for each conjugated molecule. OCH3 H2C=CH-C=N CH3 CH2 H2Carrow_forward
- o. Opioids are compounds derived from opium, which comes from the opium poppy. Although in reality, all poppies produce opoids, the drug “morphine" , codeine as well as other opioid pain killers are typically derived from opium. A quick way to find or identify these compounds is to look for a six-membered ring containing a nitrogen. Often, if you see a six-membered ring with a nitrogen, you have some sort of an opoid or a modified synthetic pain killer. With that said, morphine and codeine are typically not injected or given orally in pure form. Instead, their salts are used, such as morphine sulfate or codeine phosphate. Why are the salts used or administered instead of the pure substances (or non-salt forms)? (I am not interested in drug names, but to have you answer the why of this question.) BIU E E E E 3回fe 27 В. acen W 1 F T F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 F12 () NumLk Prt Sc Pause Brarrow_forwardSection A. Check your understanding +H' H-H 1. Explain why a carbonyl C=O bond is protonated at the lone pair while and alkene C-C bond is protonated at the pi bond. Use a MO diagram to justify your answer. &-C-Harrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond-breaking and bond-making steps. :OH: Select to Add Arrows H₂O 3 H₂ H HH H :Br:Ⓒ HH Select to Add Arrows H₂O 17 :Br: H H Please select a drawing or reagent from the question areaarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
