
Interpretation:
The major products that are formed from the given organic reaction are to be determined. The stereochemistry that exhibits by the diastereomeric products, if they are formed by these reactions is to be explained.
Concept Introduction:
The catalytic hydrogenation reaction is an addition reaction of hydrogen gas in a molecule in the presence of a catalyst.
Answer to Problem 22P
Solution:
a) The principal organic product is.:
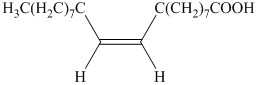
b) The principal organic product is:
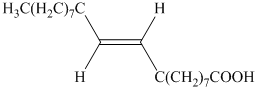
c) The principal organic product is:
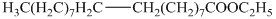
d) The principal organic product is:
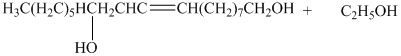
e) The principal organic product is:
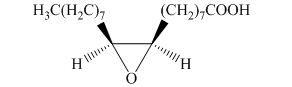
f) The principal organic product is:
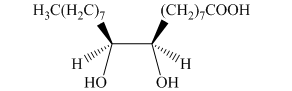
g) The principal organic product is:
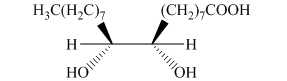
h) The principal organic product is:
i) The principal organic product is:
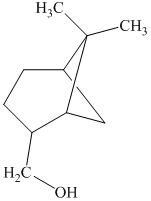
j) The principal organic product is:
Explanation of Solution
a) The given organic reaction isshown below.
The given reaction is the hydrogenation of
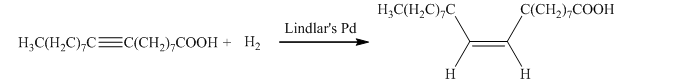
b) The given organic reaction is shown below.
The given reaction is the hydrogenation of alkynes in the presence of
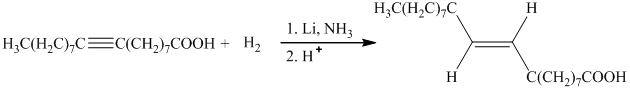
c) The given organic reaction is shown below.
The given reaction is the hydrogenation of alkenes in the presence of a platinum catalyst that reducesalkenes to
d) The given organic reaction is shown below.
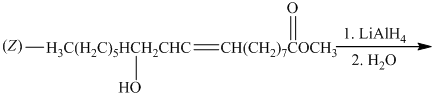
The carboxylic acid and its derivatives are reduced to the primary alcohol with the help of strong reducing agents such as lithium aluminum hydride,
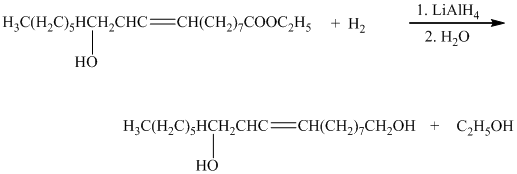
e) The given organic reaction is shown below.
In the given reaction, the epoxidation of alkene takes place. In such a reaction, the transfer of an oxygen atom from the peracid to
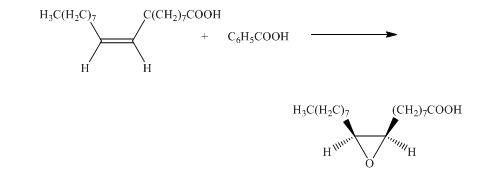
f) The given organic reaction is shown below.
In the given reaction, the hydrolysis of the product takes place that results in the breaking of the oxirane ring and forms a vicinal diol that is associated with two hydroxyl groups present on adjacent carbon atoms. The principal organic product of the given reaction is shown below.
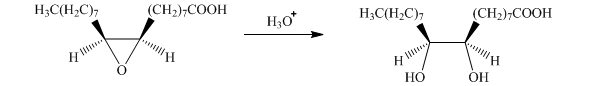
g) The given organic reaction is shown below.
In the given reaction, osmium tetraoxide
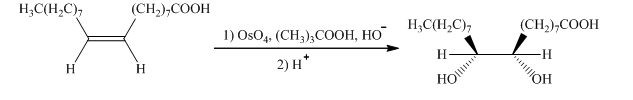
h) The given organic reaction is shown below.
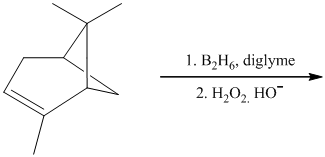
In the given reaction, hydroboration of alkenes takes place with the help of
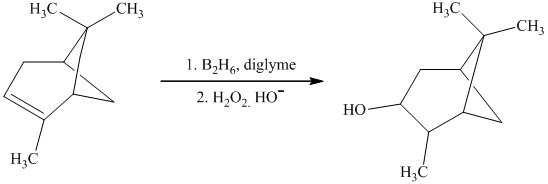
i) The given organic reaction is shown below.
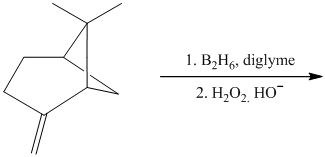
In the given reaction, hydroboration of alkenes taken place with the help of
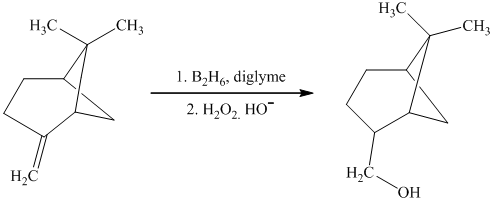
j) The given organic reaction is shown below.
In the given reaction, the dehydration of secondary alcohol is taken place in the presence of
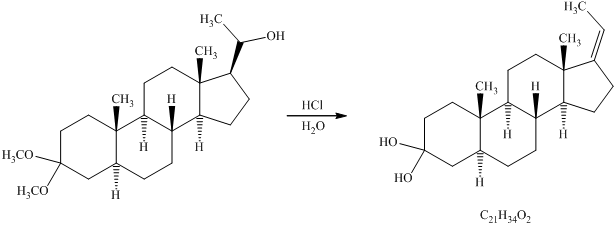
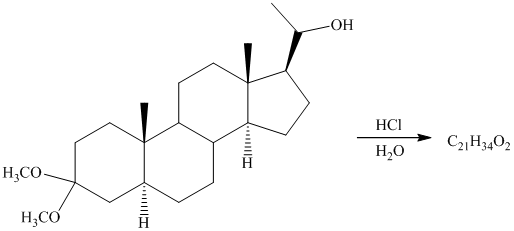
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry - Standalone book
- Draw an approximate reaction-energy diagram showing the curves for the two possible pathways for ionic addition of HBr to 1-methylcyclohexene. (a) Formation of the major product, 1-bromo-1-methylcyclohexane, and (b) formation of the minor product, 1-bromo-2-methylcyclohexane. Point out how these curves show that 1-bromo-1-methylcyclohexane should be formed fasterarrow_forwardPlease give the main substitution product for each of the following reactions, and indicate the dominant mechanism: (a) 1-bromopropane + NaOCH3 → (b) 3-bromo-3-methylpentane + NaOC2H5 →arrow_forward(a) When cis-1-bromo-2-methylcyclohexane undergoes an E2 reaction, two products (cycloalkenes) are formed. What are these two cycloalkenes, and which would you expect to be the major product? Write conformational structures showing how each is formed. (b) When rans-1-bromo-2-methylcyclohexane reacts in an E2 reaction, only one cyclo- alkene is formed. What is this product? Write conformational structures showing why it is the only product.arrow_forward
- Indicate the letter of the correct answer and kindly briefly justify the letter of answer. Cyclohexene undergoes hydrobromination. Which of these is a possible product? (A) Bromocyclohexane (B) All of these (C) Trans 1,2-dibromocyclohexane (D) Cis 1,2-dibromocyclohexanearrow_forwardDraw the structure of (2Z, 6E)-3-ethyl-7-methyldeca-2, 6-dien-1-ol, the sex pheromone of the codling moth, a common agricultural pest that destroys apple crops. By spraying an apple orchard with this pheromone, the mating of male and female moths is disrupted, and moth populations can be controlled.arrow_forwardA student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major products of formula C7H11Br. (a)Rank these three intermediates from most stable to least stable.arrow_forward
- (a) (R)-2-Bromobutane undergoes light-induced reaction with Br2 to yield a mixture of products, among which are 1,2-dibromobutane and 2,2-dibromobutane. Write the reaction showing the correct stereochemistry of the reactant. Which of the two products is optically active? Explain. ENG bj hp 17 fe 19 fho is f6 12 米 $4 4 3. & 7 23 2 8. E T Y 5 96 %24arrow_forwardWhen A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forwardThe bicyclic alkene P can be prepared by thermal electrocyclic ring closure from cyclodecadiene Q or by photochemical electrocyclic ring closure from cyclodecadiene R. Draw the structures of Q and R, and indicate the stereochemistry of the process by which each reaction occurs.arrow_forward
- Deduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of Warrow_forwardDraw a structural formula for the major organic product of the following reaction: CH3 CH3 ● CH3 CH3 AAVIL + Br₂ Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. ● If enantiomers are formed, just draw one. CH₂Cl₂ Sn [F ? ChemDoodleⓇarrow_forwardCompound A and compound B both have molecular formula C6H12. Both compounds produce epoxides when treated with a peroxy acid (RCO3H). The epoxide resulting from compound A was treated with aqueous acid (H3O+) and the resulting diol has no chiral centers. Identify the two possible structures for compound A (enter two numbers separated by commas with no spaces, e.g. "1,2") The epoxide resulting from compound B was treated with (H3O*) and the resulting diol was a meso compound. Identify compound B 2 3 potn 7 1 5 6 4 8arrow_forward
