
Interpretation:
In the structures of the given compounds, the carbon atoms that are expected to be labeled with
Concept introduction:
The isotopically labeled carbons in various compounds that are synthesized biologically enriched with
The organic material are dated using radioactive isotope of carbon
Answer to Problem 17P
Solution:
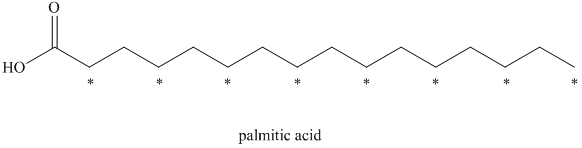
a) There are
b) There are
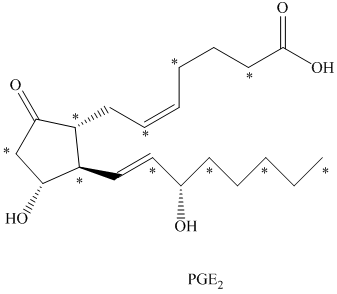
c) There are
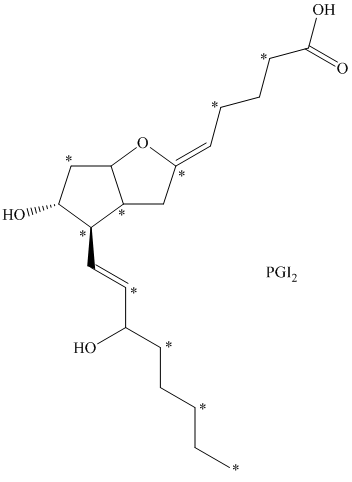
d) There are
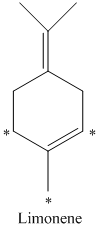
e) There are
Explanation of Solution
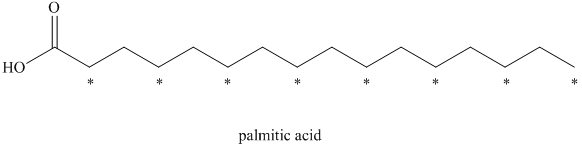
a) The given compound is Palmitic acid.
The isotopically labeled carbon atoms in palmitic acid synthesized from the acetate enriched with
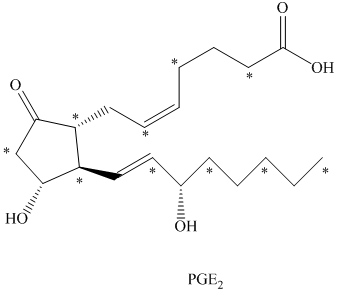
b) The given compound is
The isotopically labeled carbon atoms in
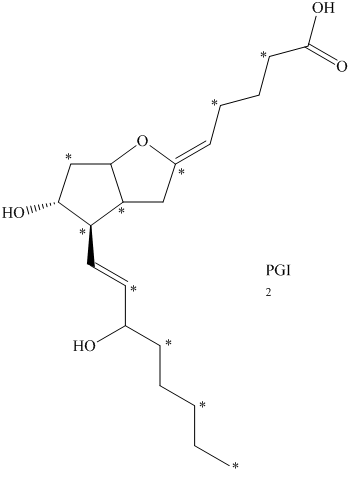
c) The given compound is
The isotopically labeled carbon atoms in
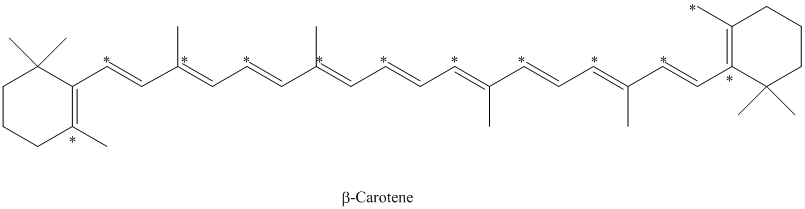
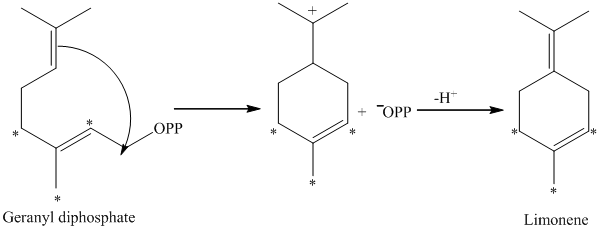
d) The given compound is limonene.
It is synthesized by the geranyl diphosphate that is further synthesized from dimethylallyl phosphate and isopentenyl diphosphate. The isotopically labeled carbon atoms in limonene enriched with
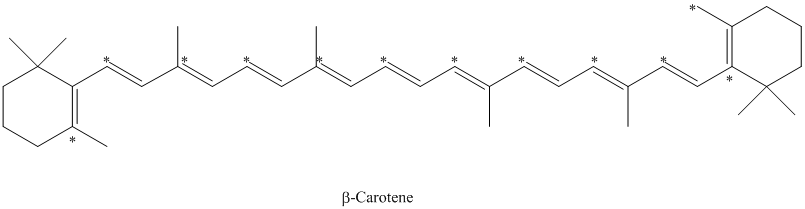
e) The given compound is
The isotopically labeled carbon atoms in
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry - Standalone book
- Draw structural formulas for the following the compounds: (a) Cis-1,3-diphenylcyclohexane (b) 5-phenylpentanoic acid (c) 3,4-dibromo-N,N-dimethylanilinearrow_forward5) Give the IUPAC name for the following structure. (a) 4-Isopropyloctane (b) 2-methyl-3-propylheptane (c) 5-Isopropyloctane (d) 2-methyl-3-ethylheptane (e) 3-ethyl-2-methylheptanearrow_forwardWhat is the structure of the alcohol produced when 3-methyl-1-pentene undergoes (a) acid catalyzed hydration (b) oxymercuration/demercuration (c) hydroboration/oxidationarrow_forward
- Write the reagent or draw structures of the starting material or organic product(s) in the following reactions. If more than one product is formed, identify the major product where possible. (a) (b) HO OH OH H2SO4 ? Cl₂ ? FeCl3arrow_forwardKetene, H2C=C=O, is an important industrial chemical. Predict the products that would be formed when ketene reacts with **hint: Markovnikov addition occurs. (a) ethanol (b) acetic acid (c) ethylamine.arrow_forwardName and draw a structural formula for the major product of each alkene addition reaction: CH S (a) CH₂C=CH₂ + HI →→→→ (b) CH 3 + HClarrow_forward
- Draw structures corresponding to the following IUPAC names: (d) 4-Ethyl-2-propyloctanoic acid (e) 2-Cyclobutenecarbonitrilearrow_forwardDraw the structures of (a) 6-methyl-3-heptanol and (b) trans-2-methylcyclohexanolarrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forward
- For each of the following molecules, please name the main functional group present: (a) O (b) O (c) OH ║ ║ │ CH3-C-CH2CH3 CH3-C- OCH3 CH3-CH2-CH2arrow_forwardWhat happens when (i) chlorobenzene is treated with Cl2/FeCl3,(ii) ethyl chloride is treated with AgNO2, and(iii) 2-bromopentane is treated with alcoholic KOH?arrow_forwardBiphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
