
a)
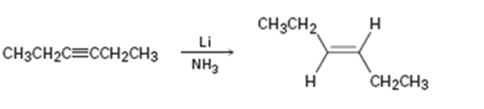
Interpretation:
Whether the mechanism of the reaction given is polar, radical or both to be identified.
Concept introduction:
A polar mechanism involves the formation of carbocation or carbanion as intermediates. A radical mechanism involves the free radicals as intermediates.
To identify:
Whether the mechanism of the reaction given is polar, radical or both.
b)
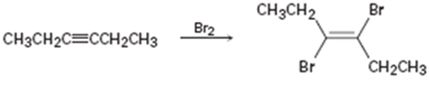
Interpretation:
Whether the mechanism of the reaction given is polar, radical or both to be identified.
Concept introduction:
A polar mechanism involves the formation of carbocation or carbanion as intermediates. A radical mechanism involves the free radicals as intermediates.
To identify:
Whether the mechanism of the reaction given is polar, radical or both.
c)
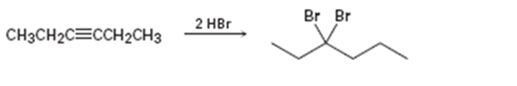
Interpretation:
Whether the mechanism of the reaction given is polar, radical or both to be identified.
Concept introduction:
A polar mechanism involves the formation of carbocation or carbanion as intermediates. A radical mechanism involves the free radicals as intermediates.
To identify:
Whether the mechanism of the reaction given is polar, radical or both.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- Alkyl halides can be reduced to alkanes by a radical reaction with tributyltin hydride, (C4H9)3SnH, in the presence of light (hv). Propose a radical chain mechanism by which the reaction might occur. The initiation step is the light-induced homolytic cleavage of the Sn-H bond to yield a tributyltin radical.arrow_forwardCompounds P and Q can undergo a Diels-Alder reaction to form two regioisomeric products R and S as shown in Figure 5. OMe O C8H12O2 R C8H12O2 S Figure 5 Draw the chemical structures of regioisomeric compounds R and S. Using possible resonance contributors of P and Q predict which of the two regioisomers will be favoured in the reaction. Using curly arrows, draw the mechanism for the reaction of P and Q to form the dominant regioisomer you have predicted in your answer to part (ii) above.arrow_forwardb) Listed below are several hypothetical nucleophilic substitution reactions. None is synthetically useful because the product indicated is not formed at an appreciable rate. In each case provide an explanation for the failure of the reaction to take place as indicated. OMe HO + OMe + OH HO + CH; OHarrow_forward
- In the presence of a radical initiator (Z•), tributyltin hydride (R3SnH, R = CH3CH2CH2CH2) reduces alkyl halides to alkanes: R′X + R3SnH → R′H + R3SnX. The mechanism consists of a radical chain process with an intermediate tin radical: This reaction has been employed in many radical cyclization reactions. Draw a stepwise mechanism for the following reaction.arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs due to a nucleophilic substitution pathway. The reaction is 100% stereospecific. Please explain what this observation tells about the mechanism of the reaction.arrow_forwardProvide the whole reaction mechanism (generation of electrophile, nucleophile, bond formation, bond breaking and movement of arrows) and the final product of the following reactions:arrow_forward
- When ethoxybenzene is treated with a mixture of nitric acid and sulfuric acid, two products are obtained, each of which has the molecular formula C8H9NO3. For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly in your products. Do not use abbreviations such as Me or Ph.arrow_forwardPredict the product(s) of the following reactions, including stereochemistry when necessary and identify the mechanism of each substitution reaction (SN1 vs SN2). Draw the reaction mechanism (reaction arrows) for any one of the reactions to show how the product is formed.arrow_forwardIllustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.arrow_forward
- When exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a chlorination reaction occurs. The products are found to contain substantial amounts of di-, tri-, and tetrachloromethane, as well as unreacted methane. Explain how a mixture is formed from this stoichiometric mixture of reactants, and propose mechanisms for the formation of these compounds from chloromethane.arrow_forwardReaction of but-1-ene with HBr gives two products in unequal amounts. In each case, identify the two products, state which is the major product, explain why it is the major product and give the mechanism for its formation.arrow_forwardDraw the mechanism, including stereochemistry, of the following reaction, predicting the products formed. What is the relationship of the products that are formed?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

