
Concept explainers
a)
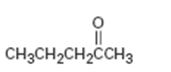
Interpretation:
The alkyne which can be used as a starting material for preparing methyl-n-propyl ketone is to be stated.
Concept introduction:
Addition of water to triple bonds occurs to yield an enol initially as the product. The enol then tautomerizes to give a ketone as the final product. Terminal
To state:
The alkyne which can be used as a starting material for preparing methyl-n-propyl ketone.
b)
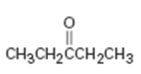
Interpretation:
The alkyne which can be used as a starting material for preparing diethyl ketone is to be stated.
Concept introduction:
Addition of water to triple bonds occurs to yield an enol initially as the product. The enol tautomerizes to give a ketone as the final product. Terminal
To state:
The alkyne which can be used as a starting material for preparing diethyl ketone.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- 4-pyranone will readily undergo an acid-base reaction. Identify the reaction conditions that will result in the formation of an aromatic product. Then, draw the aromatic resonance product structure. Include all lone pairs in your structure. Ignore inorganic byproducts. H3O+arrow_forwardHow do you produce 2-pentanol from ethyne? Draw the involved structure in the reaction.arrow_forward13. Synthesize the following compounds using materials with no more than six carbons. to H₂C OH SO3Harrow_forward
- CCH H20, H2SO4 H9SO4 CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Hjö: -CH3 -CH3 H3O*arrow_forwardSynthesize the products by drawing out reagents and intermediates along the way.arrow_forward3. Show the steps necessary, in order, to make each of the following compounds, starting with benzene. a) DE CH NO₂arrow_forward
- b) Synthesize the following product starting with phenol (hydroxybenzene). NO₂ OH HO. phenolarrow_forwardDuring multistep syntheses, reduction reactions are often performed. Functional groups react differently with these reagents. With this in mind, draw the structure of the product of this reaction: CO₂H CO₂H s HO₂C • For your convenience, the starting material has been drawn for you in the sketchpad. HO OH LIAIH4 -OH - [ ] ?arrow_forwardWhat are the steps and reagents needed to synthesize this molecule?arrow_forward
- How could the following compounds be prepared using an alkene as one of the starting materials?arrow_forwardPredict the products or starting material of the following reactions. Only the organic products are needed (no byproducts).arrow_forwardDraw structures of compounds that fit the following descriptions: a) An , unsaturated ketone, C9H8O b) An diketone c) An aromatic ketone, C9H10O d) A diene aldehyde, C7H8Oarrow_forward
