
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 27.SE, Problem 47AP
Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained:
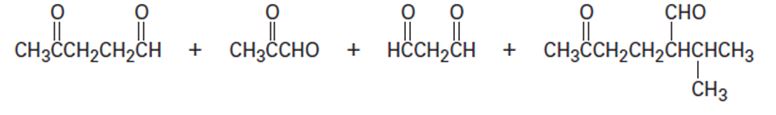
Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
α-Terpinene, C10H16, is a pleasant-smelling hydrocarbon that has been isolated from oil of marjoram. On hydrogenation over a palladium catalyst, α-terpinene reacts with 2 molar equivalents of H2 to yield a hydrocarbon, C10H20. On ozonolysis, followed by reduction with zinc and acetic acid, α-terpinene yields two products, glyoxal and 6-methyl-2,5- heptanedione.
(a) How many degrees of unsaturation does a-terpinene have?
(b) How many double bonds and how many rings does it have?
(c) Propose a structure for a-terpinene.
One compound that contributes to the “seashore smell” at beaches in Hawai‘i is dictyopterene D', a component of a brown edible seaweed called limu lipoa. Hydrogenation of dictyopterene D' with excess H2 in the presence of a Pd catalyst forms butylcycloheptane. Ozonolysis with O3 followed by (CH3)2S forms CH2(CHO)2, HCOCH2CH(CHO)2, and CH3CH2CHO. What are possible structures of dictyopterene D'?
Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16, both yield 2,6- dimethyloctane when treated with H2 and a Pd catalyst. Ozonolysis of oximene forms (CH3)2C = O, CH2 = O, CH2(CHO)2, and CH3COCHO. Ozonolysis of myrcene yields (CH3)2C = O, CH2 = O, (two equiv), and HCOCH2CH2COCHO. Identify the structures of oximene and myrcene.
Chapter 27 Solutions
Organic Chemistry
Ch. 27.1 - Carnauba wax, used in floor and furniture...Ch. 27.1 - Draw structures of glyceryl tripalmitate and...Ch. 27.2 - Prob. 3PCh. 27.2 - Write the saponication reaction of glyceryl...Ch. 27.4 - Prob. 5PCh. 27.5 - Prob. 6PCh. 27.5 - Prob. 7PCh. 27.6 - Draw the following molecules in chair...Ch. 27.6 - Lithocholic acid is an A–B cis steroid found in...Ch. 27.7 - Prob. 10P
Ch. 27.SE - Prob. 11VCCh. 27.SE - Propose a biosynthetic pathway for the...Ch. 27.SE - Identify the following fatty acid, and tell...Ch. 27.SE - Prob. 14MPCh. 27.SE - Prob. 15MPCh. 27.SE - Prob. 16MPCh. 27.SE - Prob. 17APCh. 27.SE - Fats can be either optically active or optically...Ch. 27.SE - Prob. 19APCh. 27.SE - Show the products you would expect to obtain from...Ch. 27.SE - Draw a Newman projection along the C2-C3 bond of...Ch. 27.SE - The plasmalogens are a group of lipids found in...Ch. 27.SE - Prob. 23APCh. 27.SE - Prob. 24APCh. 27.SE - Prob. 25APCh. 27.SE - Prob. 26APCh. 27.SE - Prob. 27APCh. 27.SE - Prob. 28APCh. 27.SE - Prob. 29APCh. 27.SE - Assume that acetyl CoA containing a 14C isotopic...Ch. 27.SE - Assume that acetyl CoA containing a 14C isotopic...Ch. 27.SE - Assume that acetyl CoA containing a 14C isotopic...Ch. 27.SE - Assume that acetyl CoA containing a 14C isotopic...Ch. 27.SE - Prob. 34APCh. 27.SE - Draw the most stable chair conformation of...Ch. 27.SE - Draw the most stable chair conformation of...Ch. 27.SE - Prob. 37APCh. 27.SE - Prob. 38APCh. 27.SE - Prob. 39APCh. 27.SE - What product would you obtain by reduction of...Ch. 27.SE - Prob. 41APCh. 27.SE - Eleostearic acid, C18H30O2, is a rare fatty acid...Ch. 27.SE - Prob. 43APCh. 27.SE - Prob. 44APCh. 27.SE - Propose a synthesis of diethylstilbestrol (Problem...Ch. 27.SE - Prob. 46APCh. 27.SE - Cembrene, C20H32, is a diterpenoid hydrocarbon...Ch. 27.SE - α-Fenchone is a pleasant-smelling terpenoid...Ch. 27.SE - Prob. 49APCh. 27.SE - Propose a mechanism for the biosynthesis of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Alloocimene is a hydrocarbon found in turpentine. It has the molecular formula C₁0H16 and a UV absorption maximum at 290 nm. On hydrogenation with a palladium catalyst, 2,6-dimethyloctane is obtained. Ozonolysis of alloocimene, followed by treatment with zinc and acetic acid, produces the following four fragments: || CH3C-CH i O || || CH3CCH3 HC-CH Propose a structure for alloocimene. O || CH3CHarrow_forwardIsoerythrogenic acid, C18H26O2, is an acetylic fatty acid that turns a vivid blue on exposure to UV light. On Catalytic hydrogenation over a palladium catalyst, five molar equivalents of hydrogen are absorbed, and stearic acid, CH3(CH2)16CO2H, is produced. Ozonolysis of isoerythrogenic acid yields the following products: formaldehyde, CH2O, malonic acid, HO2CCH2CO2H, adipic acid, HO2C(CH2)4CO2H, and the aldehyde carboxylic acid, OHC(CH2)6CO2H. Provide a structure for isoerythrogenic acid.arrow_forwardStructure of product C A hydrocarbon, A, with formula C11H16 undergoes hydrogenation with Pd to give product B, C11H22, and with Lindlar's catalyst to give product C, C11 H18. Ozonolysis leads to a mixture of products including cyclohexanone and 4-oxobutanoic acid. Treatment with sodium amide (NaNH2) followed by bromoethane gives compound D, C13H20. Draw possible structures for A, B, C, and D. H₂, Lindlar's Problem 13 of 14 Structure of A 1. NaNH2 Submit 2. CH3CH₂Br Structure of product Darrow_forward
- Compound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2 SO4 gives E (C10 H16), which decolorizes two equivalents of Br2 to give F (C10H16 Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to give isobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forwardTreatment of a hydrocarbon A (molecular formula C9H18) with Br2 in the presence of light forms alkyl halides B and C, both having molecular formula C9H17Br. Reaction of either B or C with KOC(CH3)3 forms compound D (C9H16) as the major product. Ozonolysis of D forms cyclohexanone and acetone. Identify the structures of A–D.arrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr) which reacts violently with D2O to give 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (Ch3COCH3) followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which reacts with 2 equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and Pt catalyst to give 2-methylpropylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughout.arrow_forward
- Hydrocarbon A has the formula C9H12 and absorbs 3 equivalents of H2 to yield B, C9H18, when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H2SO4 in the presence of mercury(II), two isomeric ketones, C and D, are produced . Oxidation of A with KMnO4 gives a mixture of acetic acid (CH3COOH) and the tricarboxylic acid E. Propose structures for compounds A-D, and write the reactions.arrow_forwardHydrocarbon A has the formula C9H12 and absorbs 3 equivalents of H2 to yield B, C9H18, when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H₂SO4 in the presence of mercury(II), two isomeric ketones, C and D, are produced. Oxidation of A with O3 gives a mixture of acetaldehyde (CH3COH) and the trialdehyde shown below. Propose structures for compounds A-D. CH,CHO OCHCH,CHCH CHOarrow_forwardCompound A (C7H11Br) is treated with magnesium in ether to give B (C7H11MgBr), which reacts violently with D2O togive 1-methylcyclohexene with a deuterium atom on the methyl group (C). Reaction of B with acetone (CH3COCH3)followed by hydrolysis gives D (C10H18O). Heating D with concentrated H2SO4 gives E (C10H16), which decolorizestwo equivalents of Br2 to give F (C10H16Br4). E undergoes hydrogenation with excess H2 and a Pt catalyst to giveisobutylcyclohexane. Determine the structures of compounds A through F, and show your reasoning throughoutarrow_forward
- An achiral hydrocarbon A of molecular formula C7H12 reacts with two equivalents of H2 in the presence of Pd-C to form CH3CH2CH2CH2CH(CH3)2. One oxidative cleavage product formed by the treatment of A with O3 is CH3COOH. Reaction of A with H2 and Lindlar catalyst forms B, and reaction of A with Na, NH3 forms C. Identify compounds A, B, and C. Be sure to answer all parts.arrow_forwardThree isomeric pentanols with unbranched carbon chains exist. Which of these isomers, upon dehydration at 180C, yields only 1-pentene as a product?arrow_forwardWhen 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY