
Concept explainers
a)
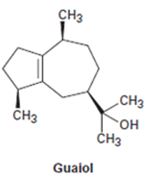
Interpretation:
The chirality centres present in Guaiol are to be indicated by asterisks and the maximum possible number of stereisomers possible is also to be stated.
Concept introduction:
A chirality centre has a carbon attached to four different groups. The maximum number of isomers possible is given by 2n where n is the number of different chiral centres present in the molecule.
To indicate:
The chirality centres present in guaiol are to be indicated by asterisks and to state the maximum possible number of stereisomers for it.
b)
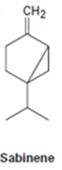
Interpretation:
The chirality centres present in sabinene are to be indicated by asterisks and the maximum possible number of stereisomers possible is also to be stated.
Concept introduction:
A chirality centre has a carbon attached to four different groups. The maximum number of isomers possible is given by 2n where n is the number of different chiral centres present in the molecule.
To indicate:
The chirality centres present in sabinene are to be indicated by asterisks and to state the maximum possible number of stereisomers for it.
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
Organic Chemistry
- in the following molecule the glycosidic bond is circled. CH2OH CH2OH H CH2OH OH HO H H НО H. ОН ÓH True Falsearrow_forwardLactose contains what number of anomeric carbons and what number of glycosidic bonds: CH2OH ОН CH2OH ОН OH OH OHarrow_forwardDraw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forward
- 17-74 Glucose, C6H12O6, contains an aldehyde group but exists predominantly in the form of the cyclic hemiacetal shown here. We will discuss this cyclic form of glucose in Chapter 20. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. (a) Which carbon in glucose provides the —OH group and which provides the —CHO group? (b) Draw the alternative chair confirmations of D-glucose and state which of the two is the more stable.arrow_forwardAssign the absolute configuration of the chiral centers of the following compounds. The number of the chiral centers of each compound is already indicated. Redraw the structures in your answer sheet. Please show your complete solution in assigning the absolute configuration of these chiral centers. он OH но, но. `NH2 CH2OH H NH2 но Ascorbic Acid Asparagine one (1) chiral center two (2) chiral centersarrow_forwardThe image shows Fischer projections for the structures of four isomers of a ketopentose. CH₂OH CH₂OH CH₂OH ПА пс C=O D HOEC H HOECH CH,OH A C=O H➡ COH но-с-н CH₂OH Which of the structures are stereoisomers of B? B Which of the structures are diastereomers of D? C=O HOI-C-H HOI-C-H CH₂OH с 000 ΤΑ B HO D CH₂OH O Which of the structures are enantiomers of C? H HI COH D CH₂OHarrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


