
Interpretation:
How to convert tropidene in to tropilidene (1,3,5-cycloheptatriene) is to be shown.
Concept introduction:
Two successive Hofmann elimination reactions of with quarternary ammonium salt of tropidene will yield tropilidene.
To show:
How to convert tropidene in to tropilidene (1,3,5-cycloheptatriene).
Answer:
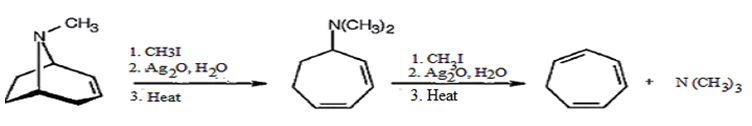
The conversion of tropidene in to tropilidene (1,3,5-cycloheptatriene) is given below.
Explanation:
Tropidene when treated with excess of methyl iodide yields a quartenary ammonium salt which undergoes Hofmann elimination when heated with Ag2O and water to produce N,N-dimethylcycloheptadiene. Methylation of the diene with excess of methyl iodide yields another quartenary ammonium salt which undergoes Hofmann elimination reaction to yield tropilidene.
Conclusion:
The conversion of tropidene in to tropilidene (1,3,5-cycloheptatriene) is given below.

Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Organic Chemistry
- 19 Which of the following ketoesters can be formed by Dieckmann cyclization? D E B C بلا سلامarrow_forwardTamoxifen is a drug used to prevent and treat breast cancer. What are all the possible metabolic reactions for this drug? Aromatic hydroxylation Epoxidation ON-dealkylation Benzylic hydroxylation O Epoxidation followed by epoxide hydrolysisarrow_forwardDraw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.arrow_forward
- Q5. What product is formed when cyclohexanone is reacted with each of the following reagents? MgBr KCN H30 H30 NABH, H30*arrow_forwardH3C a с e H3C -CH2NH2 Provide the missing reagents (a, c, e) and intermediate products (b, d) formed as toluene is converted to the final product.arrow_forwardWhat product is formed when each compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4, H2O?arrow_forward
- Prostaglandins are a class of eicosanoids, fatty acid derivatives involved in a variety of important phenomena, including fever, inflammation, and pain. The first step in prostaglandin synthesis involves the conversion of the 20-carbon fatty acid arachidonic acid to PGG2 by the enzyme prostaglandin endoperoxide synthase, or cyclooxy: Ibuprofen is one of several inhibitors of cyclooxogenase used therapeutically. Arachidonic acid 20₂ 0.5 1.0 1.5 2.5 3.5 cyclooxrygenase PGG₂ JOH محمد The following kinetic data were obtained for the enzyme cyclooxygenase in the absence of any inhibitor (1), and in the presence of ibuprofen (2) at a concentration of 48 [arachidonic acid] (uM) (1) v(uM/min) (2) v(uM/min) 23.5 32.2 36.9 41.8 44.0 16.67 25.25 30.49 37.04 38.91 Ibuprofen A. Plot the data in standard Michaelis-Menten form as well as in double-reciprocal form. B. Determine Vmax and Km for the enzyme. C. Based on this limited data set, what type of inhibition does ibuprofen likely exhibit?…arrow_forwardH3C₁ a b d e H3C Provide the missing reagents (a, c, e) and intermediate products (b, d) formed as toluene is converted to the final product. CH, NHarrow_forwardH2N Orcinol но A но он a.arrow_forward
- Which of the following is a hemiacetal? ОН ОН A) H;C-C-OH В) Н,С-С—ОСH Н н ОН нс- OCH3 D) H,C-С—ОСН, С) НС-С—ОН ÓCH3 н A Darrow_forwardB-D-Glucose, a hemiacetal, can be converted to a mixture of acetals on treatment with CH,OH in the presence of acid. Draw a stepwise mechanism for this reaction. Explain why two acetals are formed from a single starting material. он он OH CH;OH, HCI HO Но HO HO но + Но + H20 OH OCH3 OH OHOCHS но B-D-glucosearrow_forwardDraw the structure of the following organic compounds. ▪ 7-fluorohept-4-yn-2-ol 3-methoxycyclopent-3-en-1-one 5-ethyl-6-fluoro-7-propoxyhept-2-yne 5-amino-3,3-dimethylcyclohexan-1-one 3-ethoxy-N,N-diethyl-5-oxoheptanamide - ☐ ☐ ■ 1-ethoxy-3-ethyl-N-methylpent-4-en-2-araine 2-amino-5-fluorocyclopentane-1-carboxylic acid acetic 4-fluoro-3-hydroxy-2-methylpentanoic anhydridearrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

