
Concept explainers
a)
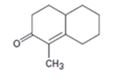
Interpretation:
How to prepare the cyclohexanone shown by combining a Stork enamine reaction with an intramolecular aldol condensation is to be shown.
Concept introduction:
The steps involved in the reaction are i) Reaction of the cyclic
To give:
How to prepare the cyclohexanone shown by combining a Stork enamine reaction with an intramolecular aldol condensation.
b)
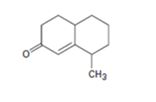
Interpretation:
How to prepare the cyclohexanone shown by combining a Stork enamine reaction with an intramolecular aldol condensation is to be shown.
Concept introduction:
The steps involved in the reaction are i) Reaction of the cyclic ketone with pyrrolidine ii) Michael addition of enamine to cyclohexanone iii) A proton transfer iv) Hydrolysis of the enamine to eliminate the amine v) Abstraction of a proton from the diketone vi) Internal aldol reaction to form the second ring vii) Protonation and dehydration.
To give:
How to prepare the cyclohexanone shown by combining a Stork enamine reaction with an intramolecular aldol condensation.
c)
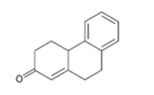
Interpretation:
How to prepare the cyclohexanone shown by combining a Stork enamine reaction with an intramolecular aldol condensation is to be shown.
Concept introduction:
The steps involved in the reaction are i) Reaction of the cyclic ketone with pyrrolidine ii) Michael addition of enamine to cyclohexanone iii) A proton transfer iv) Hydrolysis of the enamine to eliminate the amine v) Abstraction of a proton from the diketone vi) Internal aldol reaction to form the second ring vii) Protonation and dehydration.
To give:
How to prepare the cyclohexanone shown by combining a Stork enamine reaction with an intramolecular aldol condensation.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- The Favorskii reaction involves treatment of an -bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclohexanone with aqueous NaOH yields cyclopentanecarboxylic acid. Propose a mechanism.arrow_forward@GMU 73765 What is the expected product A for the following intramolecular aldol reaction? O CH3 O NaOH A H3C EtOH Harrow_forwardWhat combination of reagents can be used to make the following substance via an enamine? .&.Y.O Lay 11 B o o o o IV 8 + FO NH₂ 01 Oll II OIV O None of these reagents can be used.arrow_forward
- An acetal produces the following three compounds upon hydrolysis. Of acetals A-E which could it be? H3CO OCH3 П A B U D Acetal A H2O H₂SO4(cat.) замен поста "OCH3 В + HO. H₂CO ОН D + OCH3 CH3OH E OCH3 "OCH3arrow_forwardWhat carbonyl compound and what phosphonium ylide are needed to synthesize the following compounds?arrow_forward21.3 Aldol Reactions Predict the major product of the following reaction. NaOH MeO MeO HO H MeO. H H OMe H₂O magnam H HO MeO mayl HO H OMe OMearrow_forward
- Propose a mechanism for the followingreaction → NaOH,acetonearrow_forwardChoose the best sequence of reagents to synthesize the indicated compound.arrow_forward22. Explain, with the help of structures and curved arrow mechanisms, why trans-2-chloro- 1-cylocpentanol undergoes a Williamson ether synthesis to give an epoxide when treated with NaOH, whereas cis-2-chloro-1-cylocpentanol does not. он NaOH CI он NaOH CI No Epoxidearrow_forward
- How to synthesise 1-cyclohexylcyclohexanol from cyclohexanone?arrow_forwardThe Stork reaction is a condensation reaction between an enamine donor and an α,β-unsaturated carbonyl acceptor. The overall reaction consists of a three-step sequence of formation of an enamine from a ketone, Michael addition to an α,β-unsaturated carbonyl compound, and hydrolysis of the enamine in dilute acid to regenerate the ketone. Consider the Stork reaction between cyclohexanone and propenal Draw the structure of the product of the enamine formed between cyclohexanone and dimethylamine. - Michael addition to an α,β-unsaturated carbonyl compound, and - hydrolysis of the enamine in dilute acid to regenerate the ketone.arrow_forwardTreatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. Propose a mechanism.arrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

