
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.14, Problem 8P
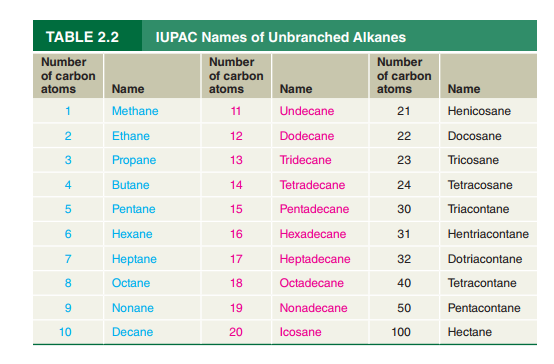
Refer to Table
Beeswax (Figure
formula for hentriacontane.
Octacosane has been found to be present in a certain fossil plant. Write a
condensed structural formula for octacosane.
What is the IUPAC name of the
the cockroach aggregation pheromone?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
College Chemistry II
CHE 132
Test #1 - Part B
1. The following names are incorrect by IUPAC rules. Draw a condensed structural formula for each
molecule listed and give the correct IUPAC name.
a) 3-propyl-4-hexyne
b) 1,4-dimethyl-5-cylcopentene
CH2 CH2 CH3
CH3-CH2
-CH- C=CH
c) 2-sec-butyl-4-pentene
d) 2,4-diethyl-2-methylpentane
2. Write the balanced equation for the complete combustion of a hydrocarbon of your choice. Name the
hydrocarbon used in the reaction.
1) Explain the similarities and differences between Lewis structures and line drawings. Provide a drawn example
different from the one shown in the video.
2) Provide a summary of the IUPAC rules. Do not copy them verbatim from the lecture video.
3) Draw and name an alkane or cycloalkane that has 3 different substituents. You can not have the same example as
someone else.
4) Draw and name an alkene that has an alkyne substituent. You can not have the same example as someone else.
5) Provide your own list of alkanes. They can not be the same structure but should all have the same number of
carbons. (see the problem solving video for an example) Rank them in order of increasing melting point. You can not
have the same example as someone else.
MacBook Air
DD
F11
F9
F10
F8
F6
F7
F5
F4
F2
F3
)
&
@
#
$
%
4
5
6
7
8
9
2
3
{
R
Y
[
+ ||
品。
1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them,
isooctane, is used as a standard in determining the octane rating of gasoline
a. Draw the structural formulas for at least ten chain isomers of octane.
b. Give the IUPAC name of each.
C. Which of the isomers that you have drawn has the highest boiling point?
Which has the lowest boiling point? Rationalize.
2. Which of the following structural formulas represent identical compounds
and which represent constitutional/structural isomers?
Identical compounds:
Constitutional isomers:
a). CH3CH2CHCH3
e). CH2CH2CHCH3
CH3
i). CH3-C-CI
ČI
CI
CI
CH3
CH2CI
b). CH3-C-CH3
f). CH3CH2CH2CH,CI
j). CICH2
CI
CH3
g). CICH,CHCH3
CH2CI
k). CH3-CH-CH3
CI
c). CH,CHCHCH3
CI
h). CH3CHCH2CH2CI
CH2CH3
1). CH3CHCI
d).
CI
CI
Chapter 2 Solutions
Organic Chemistry - Standalone book
Ch. 2.4 - Prob. 1PCh. 2.7 - Prob. 2PCh. 2.8 - Identify the orbital overlaps of all of the bonds...Ch. 2.9 - The hydrocarbon shown, called vinylacetylene, is...Ch. 2.12 - Prob. 5PCh. 2.12 - Prob. 6PCh. 2.13 - Prob. 7PCh. 2.14 - Refer to Table 2.2 as needed to answer the...Ch. 2.15 - Prob. 9PCh. 2.15 - Prob. 10P
Ch. 2.16 - Prob. 11PCh. 2.17 - Prob. 12PCh. 2.18 - Prob. 13PCh. 2.20 - Prob. 14PCh. 2.21 - Match the boiling points with the appropriate...Ch. 2.22 - Write a balanced chemical equation for the...Ch. 2.22 - Using the data in Table 2.3, estimate the heat of...Ch. 2.22 - Prob. 18PCh. 2.22 - Prob. 19PCh. 2.23 - Prob. 20PCh. 2.23 - Which of the following reactions requires an...Ch. 2 - The general molecular formula for alkanes is...Ch. 2 - Prob. 23PCh. 2 - Prob. 24PCh. 2 - Prob. 25PCh. 2 - What is the hybridization of each carbon in...Ch. 2 - Prob. 27PCh. 2 - Does the overlap of two p orbitals in the fashion...Ch. 2 - Prob. 29PCh. 2 - Aphids secrete an alarm pheromone having the...Ch. 2 - All the parts of this problem refer to the alkane...Ch. 2 - Prob. 32PCh. 2 - Prob. 33PCh. 2 - Prob. 34PCh. 2 - From among the 18 constitutional isomers of C8H18,...Ch. 2 - Give the IUPAC name for each of the following...Ch. 2 - Using the method outlined in Section 2.16, give an...Ch. 2 - Prob. 38PCh. 2 - Write a balanced chemical equation for the...Ch. 2 - The heats of combustion of methane and butane are...Ch. 2 - In each of the following groups of compounds,...Ch. 2 - Given H for the reaction H2(g)+12O2(g)H2O(l)...Ch. 2 - Prob. 43PCh. 2 - Prob. 44PCh. 2 - Prob. 45PCh. 2 - Prob. 46PCh. 2 - Prob. 47PCh. 2 - Compound A undergoes the following reactions:...Ch. 2 - Prob. 49PCh. 2 - Some Biochemical Reactions of Alkanes Alkanes...Ch. 2 - Prob. 51DSPCh. 2 - Some Biochemical Reactions of Alkanes Alkanes...Ch. 2 - Prob. 53DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part II - Isomerism 1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them, isooctane, is used as a standard in determining the octane rating of gasoline a. Draw the structural formulas for at least ten chain isomers of octane. b. Give the IUPAC name of each. c. Which of the isomers that you have drawn has the highest boiling point? Which has the lowest boiling point? Rationalize.arrow_forwardThe following compound exhibits cis/trans isomerism. Indicate for the compound: (1) the number of carbons in the longest chain, (2) the position of the double bond in the chain, (3) the IUPAC name of the compound [use the cis/trans in the name]. Answer for blank # 1: 6 Answer for blank # 2: 3 Answer for blank # 3: 3-hexenearrow_forwardPart II - Isomerism 1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them, isooctane, is used as a standard in determining the octane rating of gasoline a. Draw the structural formulas for at least ten chain isomers of octane. b. Give the IUPAC name of each. c. Which of the isomers that you have drawn has the highest boiling point? Which has the lowest boiling point? Rationalize. 2. Which of the following structural formulas represent identical compounds and which represent constitutional/structural isomers? Identical compounds: Constitutional isomers: CH3 i). CH3-C-CI ČH3 a). CH3CH2CHCH3 e). CH2CH2CHCH3 CI CI CI CH,CI b). CH3-C-CH3 f). CH3CH2CH2CH,CI j). CICH2 CI CH3 g). CICH,CHCH3 CH2CI k). CH3-CH-CH3 CI c). CH3CHCHCH3 CH2CH3 1). CH3CHCI h). CH3CHCH2CH2CI CI d). -CIarrow_forward
- Constitutional isomers are compounds which have the same molecular formula but different structural formulae. They are different compounds with different physical and chemical properties.a. Rearrange your model of n-hexane to make as many possible isomers of C6H14 as you can. Draw the structural formula and write down the IUPAC name for each isomer that you make.b. Constitutional isomers can also have different functional groups. Make all possible isomers of C3H8O. Write down the structural formulae and IUPAC names for all the compounds you make. Hint: Consider alcohol and ether functional groups.arrow_forward8. a) C3H$C1 has several structural isomers containing an alkene functional group. Complete the table by stating the systematic names and drawing the structural formulae of THREE structural isomers of C3H$Cl. Systematic Name Structural Formula b) A chemical test can be performed quickly in a school laboratory to distinguish between cyclohexanol and 1-methylcyclohexanol. Identify the reagent used in this test and describe the expected observations.arrow_forwardWhat is the difference in bonding and in general molecular formula between an alkene and a cycloalkane with the same number of carbon atoms?arrow_forward
- According to Table 2.10, five constitutional isomers are possible for alkanes with six carbon atoms. Write structural and condensed formulas for these isomers.arrow_forwardDistinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License