
Concept explainers
Write a balanced chemical equation for the combustion of each of the following compounds:
a) Decane
b) Cyclodecane
c) Methylcyclononane
d) Cyclopentylcyclopentane
Interpretation:
A balanced chemical equation for the combustion of each of the given compounds is to be written.
Concept introduction:
Alkanes are inert in acid-base reactions but undergo oxidation-reduction reactions.
During combustion, alkanes undergo oxidation.
This combustion reaction of alkanes is exothermic, and the products formed are carbon dioxide and water.
When balancing the reaction, the carbon and hydrogen is balanced first, leaving oxygen for the last to balance.
Answer to Problem 39P
Solution:
a)
b)
c)
d)
Explanation of Solution
a) Decane
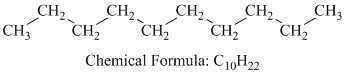
Decane is a straight chain alkane having the molecular formula
The combustion reaction of decane is written as follows:
To balance this reaction, first, the C and H are balanced.
Oxygen is balanced as follows:
The coefficients are converted to whole numbers as follows:
This is the complete balanced reaction of decane.
b) Cyclodecane
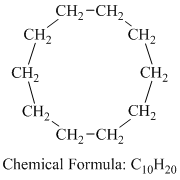
Cyclodecane is a cyclic alkane having the molecular formula
The combustion reaction of cyclodecane is written as follows:
To balance this reaction, first, the C and H are balanced.
Oxygen is balanced as follows:
This is the complete balanced reaction of cyclodecane.
c) Methylcyclononane
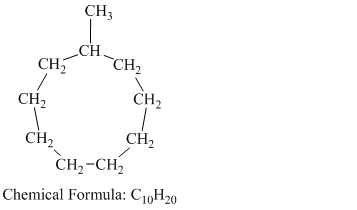
Methylcyclononane is a branched chain alkane having the molecular formula
The combustion reaction of methylcyclononane is written as follows:
To balance this reaction, first, the C and H are balanced.
Oxygen is balanced as follows:
This is the complete balanced reaction of methylcyclononane.
d) Cyclopentylcyclopentane
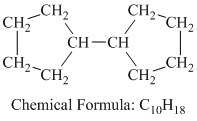
Cyclopentylcyclopentane is a branched chain alkane having the molecular formula
The combustion reaction of Cyclopentylcyclopentane is written as follows:
To balance this reaction, first, the C and H balanced.
Oxygen is balanced as follows:
The coefficients are converted to whole numbers as follows:
This is the complete balanced reaction of cyclopentylcyclopentane.
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry - Standalone book
- What is the IUPAC name for the following compound? A) hexylcyclopentane B) 1-cyclopentylhexanearrow_forwardH₂C CH3 H₂ C Name the compound below according to IUPAC rules. H H₂ H₂ pre CH3 H₂ C ·C· -C C CH₂ CH3 A) 5-methylnonane B) isodecane C) 2,2-dibutylethane D) 1,4,7-trimethylheptane E) decanearrow_forward19. A Rank the following compounds in order of increasing heat of combustion. a) nonane b) heptane d) ethane c) pentane B Which of the following will have the smallest heat of combustion? c) 2-methylpentane a) hexane b) octane d) 2,3-dimethylbutanearrow_forward
- What reactant would you use to prepare each of the following compounds from cyclohexene?arrow_forwardA semi-truck loaded with cyclohexane overturns during a rainstorm, spilling its contents over the road embankment. If the rain continues, what will be the fate of the cyclohexane?arrow_forwardWhich of the following pairs of cycloalkanes represent structural isomers? a. b. c. d.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



