
Concept explainers
What is the hybridization of each carbon in
Interpretation:
The hybridization of each carbon atom in the given molecule is to be determined along with the
Concept introduction:
A carbon atom is said to be
Multiple bonds are treated as a unit while determining the hybridization of different atoms in a molecule. For
Answer to Problem 26P
Solution:
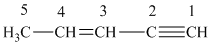
Carbon
Carbon
Carbon
Carbon
Carbon
For carbon
For carbon
For carbon
Explanation of Solution
The structure for the given compound is:
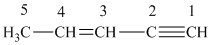
We number each carbon atom in the above compound.
A carbon atom is said to be
A carbon atom is said to be
A carbon atom is said to be
For carbon
For carbon
For carbon
The hybridization of each carbon atom in the given molecule is
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry - Standalone book
- The hybridization of the two carbon atoms differs in an acetic acid, CH3COOH, molecule. (a) Designate the correct hybridization for each carbon atom in this molecule. (b) What is the approximate bond angle around each carbon?arrow_forwardUse the VSEPR model to predict the bond angles around each central atom in the following Lewis structures (benzene rings are frequently pictured as hexagons, without the letter for the carbon atom at each vertex). Note that the drawings do not necessarily depict the bond angles correctly.arrow_forwardAspirin, or acetylsalicylic acid, has the formula C9H8O4 and the skeleton structure (a) Complete the Lewis structure and give the number of bonds and bonds in aspirin. (b) What is the hybridization about the CO2H carbon atom (colored blue)? (c) What is the hybridization about the carbon atom in the benzene-like ring that is bonded to an oxygen atom (colored red)? Also, what is the hybridization of the oxygen atom bonded to this carbon atom?arrow_forward
- Many important compounds in the chemical industry are derivatives of ethylene (C2H4). two of them are acrylonitrile and methyl methacrylate. Acrylonitrile Methyl methacrylate Complete the Lewis structures, showing all lone pairs. Give approximate values for bond angles a through f Give the hybridization of all carbon atoms. In acrylonitrile, how many of the atoms in the molecule must lie in the same plane? How many bonds and how many bonds are there in methyl methacrylate and acrylonitrile?arrow_forwardDraw the molecular orbital picture of CH3 - C= C - H. Indicate the type of hybridization on each carbon atom.arrow_forward2) Based on valence bond theory, which statement best describes the electron geometry and hybridization of the central atom(s) in acetylene HCCH? A) The electron geometry of the 2 carbons in acetylene is linear with a sp hybridization. B) The electron geometry of the 2 carbons in acetylene is trigonal planar with a sp2 hybridization. C) The electron geometry of the 2 carbons in acetylene is tetrahedral with a sp3 hybridization.arrow_forward
- What is the hybridization of the sigma bonding orbitals of the carbon atom?arrow_forwardHO: a :N CH3 -c=C-CH2 CH2 H3C Determine the hybridization and the approximate bond angles around the labeled atoms in this structure.arrow_forwardGive the type hybridization and identify the atomic molecular orbital attached to each of the carbon atoms in the given molecule. H2C = CH – CNarrow_forward
- Acrylic fibers are polymers made from a starting material called acrylonitrile, H2C(CH)CN. In acrylonitrile, a - CN group replaces a hydrogen atom on ethene. Draw the Lewis diagram for this molecule, give the hybridization of each carbon atom, and describe the \pi orbitals and the number of electrons that occupy each one. Draw the three-dimensional structure of the molecule, showing all angles.arrow_forwardWhat is the orbital hybridization of each carbon in the following: O ║ CH3–C≡C-CH2–C–CHBr-CH3arrow_forwardYeni Metin Belgesi - Not Defteri Dosya Düzen Biçim Görünüm Yardım For acetaldehyde (CH3CHO) write the Lewis structure. Write the hybridization and geometry of each carbon. Draw its 3D structure and write the bond angles (approximately).arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning


