
Concept explainers
(a)
Interpretation:
Changes in bond order, bond distance, and magnetic properties expected when
Concept Introduction:
Bond order is calculated by the expression given as follows:
The magnetic properties are related to terms such as diamagnetism and paramagnetism. Paramagnetism defines the ability of elements to be weakly attracted in an external magnetic field. It arises due to the presence of unpaired electrons. Diamagnetism defines the ability to be repelled in the external magnetic field environment. This is because diamagnetic species have paired electrons.
The bond length is estimated to be an average of covalent radii of two atoms within a bond. When bond order increases bond becomes stronger and bond length reduces. This accounts for shorter bond length in the case of unsaturated compounds while longer bonds in saturated compounds.
(a)
Explanation of Solution
For
The corresponding molecular orbitals in
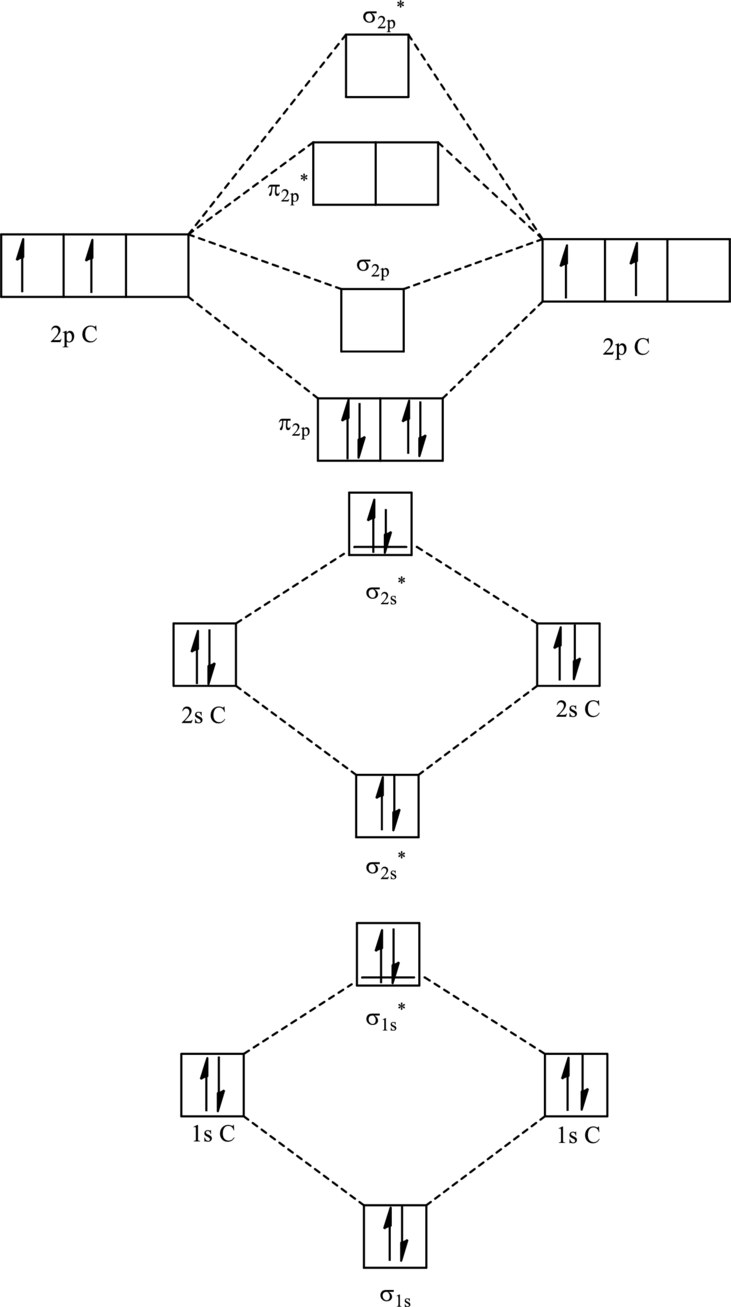
Bond order is calculated by the expression given as follows:
Substitute 4 for anti-bonding electrons and 8 for bonding electrons in equation (1).
Thus bond order is 2 in
For
Substitute 4 for anti-bonding electrons and 7 for bonding electrons in equation (1).
Thus bond order is 1.5 in
Since the reduction in bond implies bond is longer thus the bond length is more in case of
(b)
Interpretation:
Changes in bond order, bond distance, and magnetic properties expected when
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
For
The corresponding molecular orbitals in
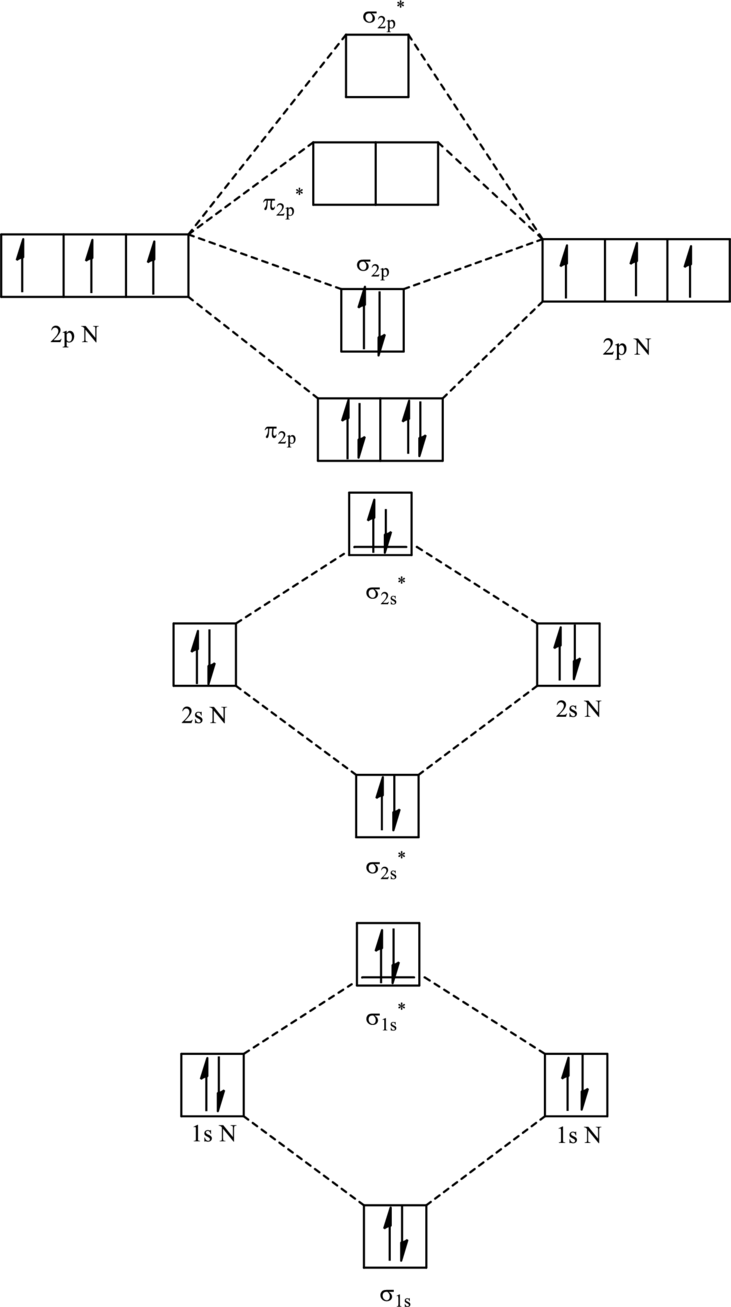
Bond order is calculated by the expression given as follows:
Substitute 4 for anti-bonding electron and 10 for bonding electrons in equation (1).
Thus bond order is 3 in
For
Substitute 4 for anti-bonding electrons and 9 for bonding electrons in equation (1).
Thus bond order is 2.5 in
Since the reduction in bond order implies bond is longer thus the bond length is more in case of
Further in
(b)
Interpretation:
Changes in bond order, bond distance, and magnetic properties expected when
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
For
The corresponding molecular orbitals in
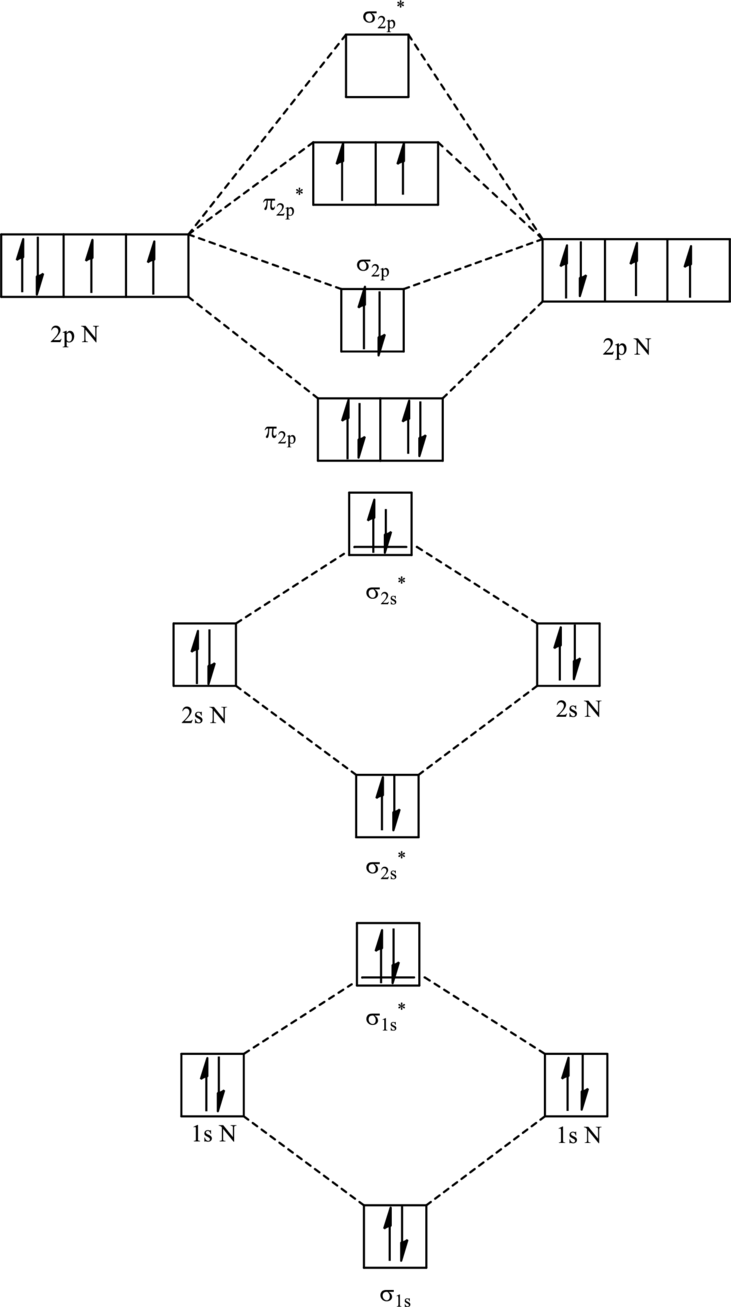
Bond order is calculated by the expression given as follows:
Substitute 6 for anti-bonding electrons and 10 for bonding electrons in equation (1).
Thus bond order is 2 in
For
Substitute 5 for anti-bonding electrons and 10 for bonding electrons in equation (1).
Thus bond order is 2.5 in
Since the reduction in bond order implies bond is shorter thus the bond length is more in case of
Further in
Want to see more full solutions like this?
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Which of the following bonds are polar: (a) P—O; (b) S—F; (c) Br—Br; (d) O—Cl? Which is the more electronegative atom in each polar bond?arrow_forwardNitrogen trifluoride (NF3) is used in the electronics industry to clean surfaces. NF3 is also a potent greenhouse gas. (A) Draw the Lewis structure of NF3 and determine its molecular geometry. (B) BF3 and NF3 both have three covalently bonded fluorine atoms around a central atom. Do they have the same dipole moment? (C) Could BF3 also behave as a greenhouse gas? Explain why or why not.arrow_forward(a) How does a polar molecule differ from a nonpolar one? (b) Atoms X and Y have different electronegativities. Will the diatomic molecule X—Y necessarily be polar? Explain. (c) What factors affect the size of the dipole moment of a diatomic molecule?arrow_forward
- a) Methane 1CH42 and the perchlorate ion 1ClO4- 2 are both described as tetrahedral. What does this indicate about their bond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar. Which of these molecules is flat?arrow_forwardFrom their Lewis structures, determine the number of sand π bonds in each of the following molecules or ions:(a) CO2; (b) cyanogen,(CN2); (c) formaldehyde, H2CO;(d) formic acid, HCOOH, which has one H and two O atomsattached to C.arrow_forwardPredict whether the bonds in the following compounds are ionic or covalent: (a) NaI (sodium iodide); (b) H 2O 2 (hydrogen peroxide).arrow_forward
- For the O2+ ion predict (a) number of unpaired electrons, (b) bond order, (c) bond enthalpy and bond length.arrow_forwardDraw the resonance forms that fit the octet rule for the metaphosphate ion, PO 3 – . (a) How many sigma bonds are there? How many pi bonds? (b) What is the phosphorus-oxygen bond order? (c) Arrange the following species in order of decreasing P-O bond strength (strongest first, weakest last). PO 4 3– , PO 2 – , PO + , PO 3arrow_forward(a) Find the angle u between adjacent nearest-neighbor bonds in the silicon lattice. Recall that each silicon atom is bonded to four of its nearest neighbors.The four neighbors form a regular tetrahedron— a pyramid whose sides and base are equilateral triangles. (b) Find the bond length, given that the atoms at the corners of the tetrahedron are 388 pm apart.arrow_forward
- Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfurtetrafluoride monoxide (OSF4) according to the followingunbalanced reaction: SF4(g) + O2(g)---->OSF4(g)The O atom and the four F atoms in OSF4 are bonded to acentral S atom. (a) Balance the equation. (b) Write a Lewisstructure of OSF4 in which the formal charges of all atomsare zero. (c) Use average bond enthalpies (Table 8.3) to estimatethe enthalpy of the reaction. Is it endothermic or exothermic?(d) Determine the electron-domain geometry ofOSF4, and write two possible molecular geometries for themolecule based on this electron-domain geometry. (e) Foreach of the molecules you drew in part (d), state how manyfluorines are equatorial and how many are axial.arrow_forwardAn important starting material for the manufacture ofpolyphosphazenes is the cyclic molecule (NPCl₂)₃. The mol-ecule has a symmetrical six-membered ring of alternating N and P atoms, with the Cl atoms bonded to the P atoms. The nitrogen-phosphorus bond length is significantly less than that expectedfor an N−P single bond.(a) Draw a likely Lewis structure for the molecule.(b) How many lone pairs of electrons do the ring atoms have?(c) What is the order of the nitrogen-phosphorus bond?arrow_forwardThe strength of a covalent bond depends upon the size of the atoms and the bond order. In general short bonds are strong bonds. For each pair of covalently bonded atoms, choose the one expected to have the higher bond energy.(A) N≡N(B) N-N ...... (A,B) fill in the blank 1(C) C=O(D) C≡O ...... (C,D) fill in the blank 2arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




