
(a)
Interpretation:
The condensed structural formula of
Concept introduction:
The condensed structural formula is a way of representation or writing organic structures in a line. The lines are drawn between bonded atoms, but the bond between carbon-hydrogen should be omitted.
Answer to Problem 18E
The condensed structural formula of
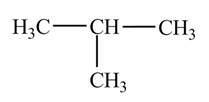
Explanation of Solution
The
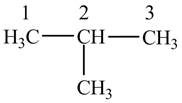
Figure 1
The numbering is assigned to the carbon atoms according to the longest continuous chain of carbon atoms. The
The condensed structural formula of
(b)
Interpretation:
The condensed structural formula of
Concept introduction:
The condensed structural formula is a way of representation or writing organic structures in a line. The lines are drawn between bonded atoms, but the bond between carbon-hydrogen should be omitted.
Answer to Problem 18E
The condensed structural formula of
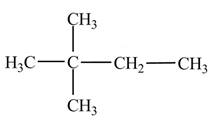
Explanation of Solution
The
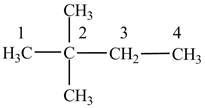
Figure 2
The numbering is assigned to the carbon atoms according to the longest continuous chain of carbon atoms. The
The condensed structural formula of
(c)
Interpretation:
The condensed structural formula of
Concept introduction:
The condensed structural formula is a way of representation or writing organic structures in a line. The lines are drawn between bonded atoms, but the bond between carbon-hydrogen should be omitted.
Answer to Problem 18E
The condensed structural formula of
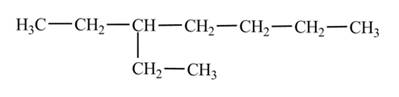
Explanation of Solution
The
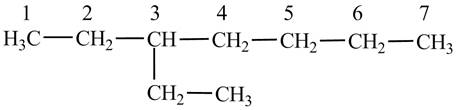
Figure 3
The numbering is assigned to the carbon atoms according to the longest continuous chain of carbon atoms. The
The condensed structural formula of
(d)
Interpretation:
The condensed structural formula of
Concept introduction:
The condensed structural formula is a way of representation or writing organic structures in a line. The lines are drawn between bonded atoms, but the bond between carbon-hydrogen should be omitted.
Answer to Problem 18E
The condensed structural formula of
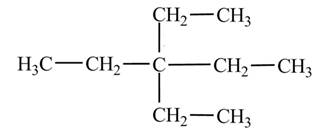
Explanation of Solution
The
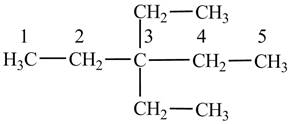
Figure 4
The numbering is assigned to the carbon atoms according to the longest continuous chain of carbon atoms. The diethyl represents that two ethyl
The condensed structural formula of
Want to see more full solutions like this?
Chapter 19 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Write structural formulas for the following compounds (includes both old- and new-style names).(a) 2-octyne (b) ethylisopentylacetylenearrow_forwardWrite the condensed formulas of the following names of organic compounds. (a). 6-ethyl-4-isopropyl-3-methyloctanearrow_forwardWrite structural formulas for the following compounds (includes both old- and new-style names).(a) 2-octyne (b) ethylisopentylacetylene (c) ethynylbenzenearrow_forward
- Write an equation for each of the following reactions. Use molecular and structural formulas and classify the reaction as combustion, addition, substitution, hydrogenation, or hydration. (a) Octane burns with oxygen gas (b) 2-methyl-1-pentene reacts with hydrogen.arrow_forwardGive an example reaction that would yield the following products. Name the organic reactant and product in each reaction. (a)alkane (b)monohalogenated alkanearrow_forwardAll of the following names are incorrect or incomplete. In each case, draw the structure (or a possible structure) and name it correctly.(a) 3-ethyl-4-methylpentane (b) 2-ethyl-3-methylpentanearrow_forward
- The following are incorrect names. Draw the compound and name it correct. (a) 1-chloro-2-bromopropane (b) 2,2-diethylbutanearrow_forwardcorrect IUPAC name of the following compounds (a) 4-methylhexane (b) 1,5-dimethyl-3-ethylpentane (c) 2,7,8-trimethyloctanearrow_forwardWrite two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.(a) 1-butanol reacts with acetic acid(b) propionic acid is poured onto solid calcium carbonatearrow_forward
- correct IUPAC name of the following compounds (a)4-methylhexane (b)1,5-dimethyl-3-ethylpentane (c)2,7,8-trimethyloctanearrow_forwardDraw astructural formula for each of the following compounds: (a)2-methylpropane (b)2,2-dimethylheptane c)2-methylpentanearrow_forward(b) Draw structures corresponding to the following IUPAC names. (a) 3,4-Dimethylnonane (c) 2,2-Dimethyl-4-propyloctane (d) 2,2,4-Trimethylpentane (b) 3-Ethyl-4,4-dimethylheptanearrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



