
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16.4, Problem 3P
Interpretation Introduction
Interpretation:
The suitable Grignard reagent that is used for the preparation of each of the given compound by taking ethylene oxide as a starting material is to be explained.
Concept introduction:
The Grignard reagent is prepared by the reaction of alkyl or aryl bromide with magnesium metal in the presence of ether.
This reagent converts the
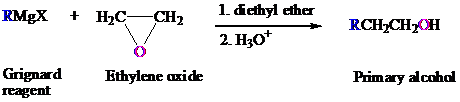
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What structural and/or molecular properties of alcohols contributes to their reactivity with oxidizing reagent?
Convert benzene into each compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.
What product can form on oxidation of a primary alcohol with an excess of oxidizing agent?
Chapter 16 Solutions
Organic Chemistry - Standalone book
Ch. 16.2 - Prob. 1PCh. 16.2 - Prob. 2PCh. 16.4 - Prob. 3PCh. 16.5 - Prob. 4PCh. 16.5 - Give the structures, including stereochemistry,...Ch. 16.5 - Prob. 6PCh. 16.7 - Prob. 7PCh. 16.8 - Prob. 8PCh. 16.8 - Prob. 9PCh. 16.9 - Predict the principal organic product of each of...
Ch. 16.9 - Prob. 11PCh. 16.10 - Prob. 12PCh. 16.11 - Prob. 13PCh. 16.12 - Prob. 14PCh. 16.12 - Prob. 15PCh. 16 - Write chemical equations, showing all necessary...Ch. 16 - Write chemical equations, showing all necessary...Ch. 16 - Which of the isomeric C5H12O alcohols can be...Ch. 16 - Prob. 19PCh. 16 - Write equations showing how 1-phenylethanol could...Ch. 16 - Write equations showing how 2-phenylethanol could...Ch. 16 - Prob. 22PCh. 16 - Show how each of the following compounds can be...Ch. 16 - Prob. 24PCh. 16 - Write the structure of the principal organic...Ch. 16 - Prob. 26PCh. 16 - Prob. 27PCh. 16 - Prob. 28PCh. 16 - Prob. 29PCh. 16 - Prob. 30PCh. 16 - (a) The cis isomer of 3-hexen-1-ol...Ch. 16 - Prob. 32PCh. 16 - Complete each of the following equations by...Ch. 16 - Prob. 34PCh. 16 - Prob. 35PCh. 16 - A diol (C8H18O2) does not react with periodic...Ch. 16 - Identify the compound C8H10O on the basis of its...Ch. 16 - dentify each of the following C4H10O isomers on...Ch. 16 - Prob. 39PCh. 16 - A compound C6H14O has the spectrum shown in Figure...Ch. 16 - Prob. 41DSPCh. 16 - Prob. 42DSPCh. 16 - Prob. 43DSPCh. 16 - Prob. 44DSPCh. 16 - Prob. 45DSPCh. 16 - Prob. 46DSPCh. 16 - Prob. 47DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Aldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forward1. Determine the structure of Grignard reagent and carbonyl compound as the starting materials to produce the following alcohols. a) он b) CH2OH c) OH d) Lonarrow_forwardGive the condensed formula of the Grignard reagent and carbonyl compound you will use to prepare the following alcohols. List all possibilities. 2-methyl propan-2-ol 3-phenyl pentan-3-ol 1-ethyl cyclohexanol 1-cyclopentyl 2-propanolarrow_forward
- give any two methods by which we can prepare ethers?arrow_forwardAccording to Le Chatelier's Principle, acetal product formation tends to INCREASE (rather than decrease) when more alcohol is added to the reaction mixture. True or False?arrow_forwardIdentify the best reagents to complete the following reaction.arrow_forward
- Which of the following compounds does not form an alcohol when it reacts with excess Grignard reagent?arrow_forwardDescribe a chemical procedure to separate a mixture of benzyl alcohol and o-cresol and to recover each in pure form. CH,OH CH3 Benzyl alcohol o-Cresolarrow_forwardDibenzalpropanone is a compound that can absorb UV rays and can be used as a sunscreen. Write down the reagents used to synthesize the compound dibenzalpropanone.arrow_forward
- Compounds A, B, and C are subjected to the Lucas test, and an oily layer was formed almost instantly after the addition of reagents to Compound C. What could be compound C? CHoices: Ethanol Isopropyl Alcohol tert-butyl alcoholarrow_forwardWhich of the following compounds will give a secondary alcohol when treated with Grignard reagent? A НСНО В C6H5COCH3 C6H5COC6H5 CH3CH2CH2CHOarrow_forwardReaction of ethylmagnesium bromide with which of the following compounds yields a secondary alcohol after quenching with aqueous acid? H₂CO CH3CHO (CH3)2CO ethylene oxide On-butyllithiumarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning