
Concept explainers
dentify each of the following
NMR spectra:
Interpretation:
The structures of each of the
Concept introduction:
In
The number of signals in a spectrum gives information about the type of carbon atoms present in the structure of the compound.
In
The
Index of hydrogen deficiency (IHD) is calculated by the equation as follows:
Here,
Oxygen atoms do not disturb the index of hydrogen deficiency.
At
At
A methine is
Answer to Problem 38P
Solution: The structures of each of the
a)
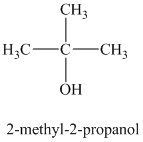
b)
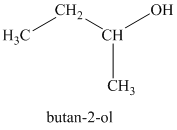
c)
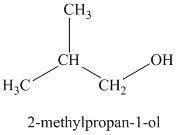
Explanation of Solution
a)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the alcohol contains no ring or multiple bonds. The
The signal
The signal
The structure must contain three equivalent methyl groups and one tertiary carbon atom. Therefore, the structure of this isomer is shown as follows:
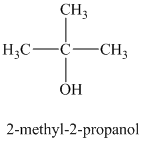
This isomer is a tertiary alcohol.
b)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the alcohol contains no ring or multiple bonds. The
The signal
The signal
The signal
Thus, the structure must contain two non-equivalent methyl groups, one methylene group, and one methine group to which the hydroxyl group is attached.
Therefore, the structure of this isomer is shown as follows:

This isomer is a secondary alcohol.
c)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the alcohol contains no ring or multiple bonds. The
The signal
The signal
The signal
Thus, the structure must contain two equivalent methyl groups, one methylene group, and one methine group to which the hydroxyl group is attached.
Therefore, the structure of this isomer is shown as follows:

Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry - Standalone book
- An unknown compound, C4H10O gave the following proton NMR data: Singlet at 3.32 ppm (3H) Triplet at 0.94 ppm (3H) Triplet at 3.35 ppm (2H) Multiplet 1.47 ppm (2H) What is the structure of the compound?arrow_forwardAn unknown compound has the molecular formula C9H10O and the Carbon-13 NMR spectrum shown below. Using this information, draw the structure of the unknown compoundarrow_forwardThe H1H1 NMR spectrum shown corresponds to an unknown compound with the molecular formula C6H10C6H10. There are no strong IR bands between 2100 and 2300 or 3250 and 3350 cm−1. Deduce and draw the structure of the molecule that corresponds to the spectrum.arrow_forward
- The 'H NMR spectrum of compound A (C3H100) has four signals: a multiplet at 8 = 7.25-7.32 ppm (5 H), a singlet at d = 5.17 ppm (1 H), a quartet at d = 4.98 ppm (1 H), and a doublet at ô = 1.49 ppm (3 H). There are 6 signals in its 13C NMR spectrum. The IR spectrum has a broad absorption in the -3200 cm-1 region. Compound A reacts with KMNO4 in a basic solution followed by acidification to give compound B with the molecular formula C7H6O2. Draw structures for compounds A and B.arrow_forward1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardCompound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward
- 07) The proton NMR spectrum of a compound with formula C5H12O2 is shown below. THE The carbon-13 NMR spectrum has three peaks, respectively at 15 ppm, 63 ppm and 95 ppm. Based on this information draw the structure of this compound.arrow_forwardAssume that you have a compound with the formula C4H8O. a) How many double bonds and/or rings does your compound contain? b) If your compound shows an infrared absorption peak at 1715 cm-1, what functional group does it have? c) If your compound shows a single 1H NMR absorption peak at 2.1 δ, what is its structure?arrow_forwardThe H NMR spectra corresponds to a hydrocarbon with the molecular formula C6H₁4. Deduce the structure from the spectra. Draw the unknown compound. 'H NMR 12 H Select Draw Rings More 1.5 1.4 2 H 1.5 1.0 0.5 0.0 300-MHz H NMR spectrum ppm (8) 0.9 0.8 (CH3)4Si G G Q2Earrow_forward
- Compound 2 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Provide structure for compound 2, explain how you reached your conclusion.arrow_forwardHow many peaks will the following molecule show in its ¹H NMR spectrum? 1 2 IN 3 6 H3C CH3 methyl acetatearrow_forwardA compound with the molecular formula C4H₁1N has the following ¹H NMR spectrum, in which the relative integration values are illustrated with step curves. Which of the following is the correct number of protons giving rise to each signal? al O2H, 3H, 6H Ο 1Η, 4Η, 6Η O 2H, 4H, 5H O 3H, 4H, 5H PPMarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
