
Concept explainers
(a)
Interpretation:
A mole concept map is to be drawn and the grams of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 20E
The mole concept map is shown below.
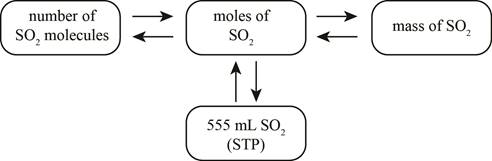
The grams of
Explanation of Solution
When
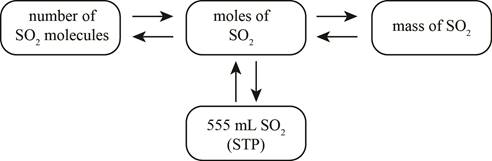
Figure 1
The volume occupied by
The volume of dissolved
The relation between
The probable unit factors are given below.
The unit factor to determine
Therefore, the volume in
Therefore, the number of moles which occupy
The molar mass of
Therefore, the mass of
The formula to calculate the mass of
Substitute the mass of
Therefore, the grams of
The grams of
(b)
Interpretation:
A mole concept map is to be drawn and the molecules of
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 20E
The mole concept map is shown below.
The molecules of
Explanation of Solution
When
Figure 1
The volume occupied by
The volume of dissolved
The relation between
The probable unit factors are given below.
The unit factor to determine
Therefore, the volume in
Therefore, the number of moles which occupy
The molecules present in
The formula to calculate the molecules occupied by
Substitute the molecules in
Therefore, the molecules of
The molecules of
(c)
Interpretation:
A mole concept map is to be drawn and the molar concentration of the sulfuric acid solution when
Concept introduction:
A mole is a basic unit used in the International system of units (SI). It is abbreviated as
Answer to Problem 20E
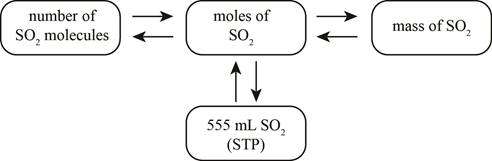
The mole concept map is shown below.
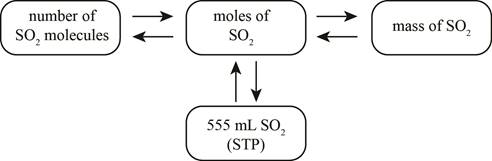
The molar concentration of the sulfuric acid solution is
Explanation of Solution
When
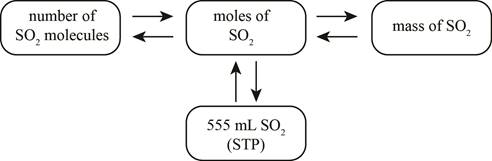
Figure 1
The volume occupied by
The volume of dissolved
The relation between
The probable unit factors are given below.
The unit factor to determine
Therefore, the volume in
Therefore, the number of moles which occupy
The number of moles in
The formula to determine molarity is shown below.
Where
•
•
•
Substitute the value of number of moles and volume in equation (1).
The relation between
The unit factors are given below.
The unit factor to determine
Therefore,
Therefore, the molar concentration of
The molar concentration of sulfurous acid solution is
Want to see more full solutions like this?
Chapter 15 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Mixtures of gases are always true solutions. True or false? Explain why.arrow_forwardWhen calcium carbonate is heated strongly, it evolves carbon dioxide gas. CaCO3(s)CaO(s)+CO2(g) 25 g of CaCO3 is heated, what mass of CO2would be produced? What volume would this quantity of CO2 (CU at STP?arrow_forwardRefer to the oxygen concentration example in Sec. 1.7.2.(a) Given that nitrogen is lighter in weight than oxygen, is N2 concentration at 10 kmmore or less that 25% of the sea level N2 concentration?(b) What is the ratio of N2 concentration to O2 at 10 km? At sea level, the ratio is 4 to 1.arrow_forward
- 205 L of carbon dioxide gas at STP is compressed and then dissolved in 10.0L of water. What is the molar concentration of the resulting solution ?arrow_forwardCalculate the new molarity if each of the following dilutions is made. Assume the volumes are additive. (a) 52.3 mL of water is added to 23.3 mL of 0.123 M CaCl2 solution M(b) 106 mL of water is added to 53.1 mL of 0.827 M KOH solution M(c) 640. mL of water is added to 151 mL of 33.0 M NaCl solution M(d) 365 mL of water is added to 724 mL of 1.92 M NaOH solutionarrow_forwardWhat volume of 0.350 M HNO3 Can be made from 150mL of 6.6 M HNO3? L (Give answer to correct sig figs in the form of units of L)arrow_forward
- Suppose that 0.3744 L of 1.056 mol / L of barium chloride is added to 0.5744 L of 1.356 mol / L of sodium sulfate (excess). Barium chloride reacts with sodium sulfate as per the equation above. To calculate the number of moles of barium chloride which are present initally before the reaction, you should use the following formula: n = c x v The number of mole value is:arrow_forwardIf 12.5 g of Glucose is dissolved in enough water to make 750 ml solution. (1) What is the molarity of the solution?arrow_forwardWhat percent of water is in the compound Magnese (ii) chloride tetrahydrate?arrow_forward
- Determine the molarity for each of the following solutions: (d) 10.5 kg of Na2SO4·10H2O in 18.60 L of solutionarrow_forwardWhat is the volume of 0.25mol of CO2 gas at STParrow_forwardWhich of the following refers to the amount of solute present in a given quantity of solvent or solution? Group of answer choices Saturation Concentration Dilution Supersaturationarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

