
a)
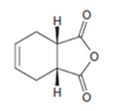
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
b)
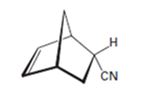
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
c)
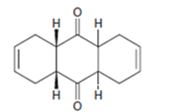
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
d)
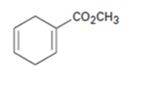
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
- Show the product of the Diels–Alder reaction of the following diene with 3-buten-2-one, H2C = CHCOCH3. Make sure you show the full stereochemistry of the reaction product.arrow_forwardBicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forwardWhat descriptive term is applied to the type of diene represented by 2,4-hexadiene? O isolated diene O cumulated diene O conjugated diene O alkynyl diene O saturated dienearrow_forward
- Which diene and dienophile would react to give the following Diels-Alder product? CN CNarrow_forwardChlordane, like DDT, is an alkyl halide that was used as an insecticide for crops such as corn and citrus and for lawns. In 1983, it was banned for all uses except against termites, and in 1988, it was banned for use against termites as well. Chlordane can be synthesized from two reactants in one step. One of the reactants is hexachlorocyclopentadiene. What is the other reactant?arrow_forwardDraw the products of the following Diels–Alder reactions. Indicate stereochemistry where appropriate.arrow_forward
- What descriptive term is applied to the type of diene represented by allene (1,2-propadiene)? O isolated diene O conjugated diene O cumulated diene O alkynyl diene O saturated diene DELarrow_forwardDraw the products of each reaction. Indicate the stereochemistry of Diels–Alder products.arrow_forwardWhat are the starting reactants for the following reaction? 4 X = cyclohexa-1,3-diene; Y = ethyl acrylate X = cyclopenta-1,3-diene; Y = acrylaldehyde X = cyclohexa-1,3-ene; Y = ethyl acrylate X = hexa-1,3-diene; Y = ethyl propiolate CO₂C₂H5arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

