
a)
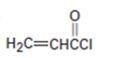
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
b)
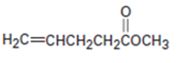
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
c)
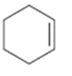
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
d)
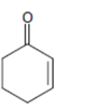
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
e)
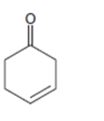
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
- Which diene is more reactive in a Diels–Alder reaction? CH2=CHCH=CHOCH3 or CH2=CHCH=CHCH2OCH3arrow_forwardDraw the products of the following Diels–Alder reactions. Indicate stereochemistry where appropriate.arrow_forwardHow many linear dienes have molecular formula C6H10? (Disregard cis–trans isomers.) A. How many of the linear dienes in part a are conjugated dienes? B. How many are isolated dienes? C. How many are cumulated dienes?arrow_forward
- Rank the following dienes in their rate of reaction with the same dienophile in a Diels-Alder reaction. For example, 1 = fastest or most reactive diene, 4 = slowest or least reactive diene.arrow_forward23. Draw all possible Diels-Alder products (show stereochemistry) and determine which is the major product. OCH, wwwww CNarrow_forward(a) Draw the major products A, B, and C for the Diels-Alder reactions below. A EtO2C. + В `CO̟Etarrow_forward
- The bicyclic alkene P can be prepared by thermal electrocyclic ring closure from cyclodecadiene Q or by photochemical electrocyclic ring closure from cyclodecadiene R. Draw the structures of Q and R, and indicate the stereochemistry of the process by which each reaction occurs.arrow_forwardDraw the products of the following Diels-Alder Reactions. Remember to show the correct stereochemistry! CH3 1. coocH3 2.arrow_forwardm. SN1 SN2 E1 E2 CH3CH2CH2CH2-OH + POCI3 with pyridine Product(s) of reaction m.arrow_forward
- Determine the double bond stereochemistry (E or Z) for the following molecules. F. H3C C A CH3 F. H B D Brarrow_forwardGive the molecule that undergoes an intramolecular Diels- Alder cycloaddition to produce the given cycloadduct product.racemic H .OCH 3 racemicarrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + HCI My 3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH₂ CHOCH3 TT CI CH3 L ▼ {n [F ? ChemDoodleⓇarrow_forward
