
The following data resulted from an experiment to assess the potential of unburnt colliery spoil as a medium for plant growth. The variables are x 5 acid extractable cations and y 5 exchangeable acidity/total cation exchange capacity (“Exchangeable Acidity in Unburnt Colliery Spoil,” Nature, 1969: 161):
| x | -5 | 16 | 26 | 30 | 38 | 52 | |
| y | 1.50 | 1.46 | 1.32 | 1.17 | .96 | .78 | .77 |
| x | 58 | 67 | 81 | 96 | 100 | 113 | |
| y | .91 | .78 | .69 | .52 | .48 | .55 |
Standardizing the independent variable x to obtain
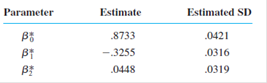
a. Estimate
b. Compute the value of the coefficient of multiple determination. (See Exercise 28(c).)
c. What is the estimated regression function
d. What is the estimated standard deviation of
e. Carry out a test using the standardized estimates to decide whether the quadratic term should be retained in the model. Repeat using the unstandardized estimates. Do your conclusions differ?
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Probability and Statistics for Engineering and the Sciences
- Find the intensities of earthquakes whose magnitudes are (a) R=6.0 and (b) R=7.9.arrow_forwardThe following fictitious table shows kryptonite price, in dollar per gram, t years after 2006. t= Years since 2006 0 1 2 3 4 5 6 7 8 9 10 K= Price 56 51 50 55 58 52 45 43 44 48 51 Make a quartic model of these data. Round the regression parameters to two decimal places.arrow_forwardAn owner of a health spa studied the relationship between “temperature in degrees Fahrenheit at 11 A.M.” (X) and “number of customers using the facilities at that time” (Y) during the summer. The data are shown below: X Y 65 18 67 18 75 22 81 19 85 27 87 24 MX=76.67 SDX=8.44 ZX ZY -1.38 -1.15 -0.20 +0.51 +0.99 +1.22 Which of the following statistics has the highest mathematical value? Standard deviation of Y Variance of Y Median of Y Mean of Yarrow_forward
- 18.2 Well water having a total hardness of 300 mg / L as CaCO , is softened by split treatment . A zeolite bed takes 3/4 of the total flow and produces water at a hardness of 3 mg / L . as CaCO , One - fourth of flow is directly mixed with the softened water . The zeolite bed has 28.3 m² resin capacity . The water softening capacity of the resin is 62 kg / m³ total hardness as CaCO ,. Exhausted resin is regenerated by 98 percent pure NaCl solution . The salt consumption is three times the theoretical amount and used as 4 percent salt solution . Calculate ( a ) regeneration period if 950 m³ per day of water is passed through the softener , ( b ) the hardness of the fin ishod water , and ( c ) salt consumption per regeneration cyclearrow_forwardpan's high population density has resulted in a multitude of resource-usage problems. One especially serious difficulty concerns waste removal. An article reported the development of a new compression machine for processing sewage sludge. An important part of the investigation involved relating the moisture content of compressed pellets (y, in %) to the machine's filtration rate (x, in kg-DS/m/hr). The following data was read from a graph in the article. x 125.8 98.1 201.4 147.3 145.9 124.7 112.2 120.2 161.2 178.9 159.5 145.8 75.1 151.5 144.2 125.0 198.8 133.9 y 77.9 76.8 81.5 79.8 78.2 78.3 77.5 77.0 80.1 80.2 79.9 79.0 76.9 78.2 79.5 78.1 81.5 71.0 (a) Determine the slope and intercept of the estimated regression line. (Round your answers to 5 decimal places, if needed.)slope: intercept: (b) Does there appear to be a useful linear relationship? Carry out a test using the ANOVA approach and a significance level of 0.05. State the appropriate null and alternative hypotheses.…arrow_forwardConsider the following data relating hours spent studying (X) and average grade on course quizzes (Y): X Y 5 6 3 8 4 8 7 10 5 7 6 9 Compute: ExEy/n 1140 6 240 235arrow_forward
- An article in the Journal of the American Ceramic Society, "Rapid Hot-Pressing of Ultrafıne PSZ Powders" (1991, Vol. 74, pp. 1547-1553) considered the microstructure of the ultrafıne powder of partially stabilized zirconia as a function of temperature. The data are shown below: x= Temperature (°C) 1100 1200 1300 1100 1500 1200 1300 y = Porosity (%) 30.8 11.4 7.7 19.2 6 13.5 3.6 (a) Fit the simple linear regression model using the method of least squares. Find the least squares estimates of the intercept and slope in the simple linear regression model. Find an estimate of o. i (Round your answer to 3 decimal places.) (Round your answer to 5 decimal places.) 6 = i (Round your answer to 2 decimal places.) (b) Estimate the mean porosity for a temperature of 1447 °C. ŷ = (Round your answer to 2 decimal places.)arrow_forwardAmericium 241 (241Am) is a radioactive material used in the manufacture of smoke detectors. The article "Retention and Dosimetry of Injected 241Am in Beagles"t described a study in which 55 beagles were injected with a dose of 241Am (proportional to each animal's weight). Skeletal retention of 241 Am (in microcuries per kilogram) was recorded for each beagle, resulting in the following data. 0.196 0.451 0.498 0.411 0.324 0.190 0.489 0.300 0.346 0.448 0.188 0.399 0.305 0.304 0.287 0.243 0.334 0.299 0.292 0.419 0.236 0.315 0.447 0.585 0.291 0.186 0.393 0.419 0.335 0.332 0.292 0.375 0.349 0.324 0.301 0.33 0.408 0.399 0.303 0.318 0.468 0.441 0.306 0.367 0.345 0.428 0.345 0.412 0.337 0.353 0.357 0.320 0.354 0.361 0.329 A USE SALT (a) Construct a frequency distribution for these data. Commute Time Frequency 0.15 to <0.20 10 0.20 to <0.25 11 0.25 to <0.30 48 0.30 to <0.35 0.35 to <0.40 0.40 to <0.45 0.45 to <0.50 0.50 to <0.55 0.55 to <0.60arrow_forward
- Algebra & Trigonometry with Analytic GeometryAlgebraISBN:9781133382119Author:SwokowskiPublisher:Cengage
 Calculus For The Life SciencesCalculusISBN:9780321964038Author:GREENWELL, Raymond N., RITCHEY, Nathan P., Lial, Margaret L.Publisher:Pearson Addison Wesley,
Calculus For The Life SciencesCalculusISBN:9780321964038Author:GREENWELL, Raymond N., RITCHEY, Nathan P., Lial, Margaret L.Publisher:Pearson Addison Wesley, Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning  Algebra: Structure And Method, Book 1AlgebraISBN:9780395977224Author:Richard G. Brown, Mary P. Dolciani, Robert H. Sorgenfrey, William L. ColePublisher:McDougal Littell
Algebra: Structure And Method, Book 1AlgebraISBN:9780395977224Author:Richard G. Brown, Mary P. Dolciani, Robert H. Sorgenfrey, William L. ColePublisher:McDougal Littell




