
Concept explainers
(a)
Interpretation:
The IUPAC name for the following alkene should be determined:
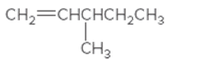
Concept Introduction:
(b)
Interpretation:
The IUPAC name for the following alkene should be determined:
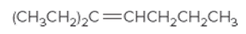
Concept Introduction:
Alkenes are named in the same way as alkanes, but the alkenes are identified by the suffix −ene, replaced instead of the ending of the name of the parent alkane. The longest carbon chain should be numbered in a way that gives the double bond the lower number. Then the compound should be named using the first number assigned to the double bond. The names of the substituents should be written first in the alphabetic order with their position on the chain.
(c)
Interpretation:
The IUPAC name for the following alkene should be determined:
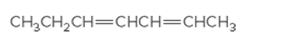
Concept Introduction:
Alkenes are named in the same way as alkanes, but the alkenes are identified by the suffix −ene, replaced instead of the ending of the name of the parent alkane. The longest carbon chain should be numbered in a way that gives the double bond the lower number. Then the compound should be named using the first number assigned to the double bond. The names of the substituents should be written first in the alphabetic order with their position on the chain.
(d)
Interpretation:
The IUPAC name for the following alkene should be determined:
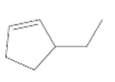
Concept Introduction:
In the nomenclature of cycloalkenes, the double bond is always located between C-1 and C-2. And 1 is not mentioned in the name. the ring is then numbered in a way that the first substituent getting the lowest number.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- I. Write the IUPAC name of each organic compound. Structure NH2-C-CH-C СН 2-О-СН2-СH3 ČI ČI 1. H3C-CH=CH-C-CH, 2. OH CH CHCH CC=CH CH CH 3.arrow_forwardWhich structure below represents 4-methylpent-2-ene? a. CH3-CH2-CH2-CH2-C=CH2 O b. CH3-CH2-C=CH-CH3 O c. O d. CH2-CH3 CH3 CH3-CH-CH=CH-CH3 CH3 CH3-CH2-CH-CH=CH2 CH, CH3arrow_forward17. Name the following alkynes. b. a H HーC三c- H-c=c- H H H. H d. H-cEc-CH,-CH3 -c=c- H- с—н H H .CH3 f. H2Ç .CH2 c-c=CH H3C. H H-C-H CH H-C C-C- -c- C-H CH3 H2C. H H Н-с-н CH2 H CH3 I-O- O-arrow_forward
- 12) Which structure is different from the compound (I) ? CH₂ CH₂- H H a H CH(CH3)2 H. H CH₂ CH₂ H b -CH(CH₂)2 CH₂ CH₂- H H C H CH(CH₂)2 H7 CH₂ CH₂ CH(CH3)2 darrow_forwardWhich structure is trans-3-hexene? CH3. H CH3, H OCH3CH₂ H OCH3CH₂ H C. C. FC. C H CH₂CH₂CH3 CH₂CH₂CH3 H H CH₂CH3 CH₂CH3 Harrow_forwardName each alkene. a. CH;-CH,-CH=CH-CH,-CH; b. CH,-CH-CH=CH-CH, CH3 CH; CH, c. CH,-CH-CH=C-CH-CH, CH3 CH; CH, d. CH;-C-CH=C-CH2-CH, CH3arrow_forward
- Complete and balance each hydrocarbon combustion reaction. a. CH;CH;CH,CH3 + O2 b. CH2=CHCH, + 02 c. CH=CCH,CH3 + O2arrow_forwardStructure A Cis Isomer H₂C Trans Isomer Name of the Alkene H C=C CH₂CH₂ H [Choose ] [Choose ] Pentene Propene Hexene Structure A Structure B [Choose ] H₂C H C=C Structure B I CH₂CH₂arrow_forwardQuestion 9: Draw the products formed when each alkyne is treated with O3 followed by H₂O. 5arrow_forward
- 8. Give the IUPAC name for each alkene. CH3 CH2CH,CH3 Br а.arrow_forwardCHCHS CH, CH CH CHCH, A D Which is the most stable alkene? Choose. Which is the least stable alkene? Choose.arrow_forwardGive the common name and IUPAC name: 4. (CH3)3CCH2COBr 5. C3H7CONHCH3 6. C-C-C-C-C-C-COCI C-C-C C | Carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





