
Concept explainers
What
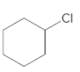
a.
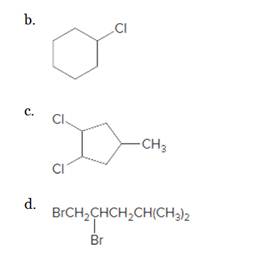
(a)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
Starting alkene-
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction:
(b)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
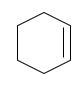
Starting alkene −
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction;
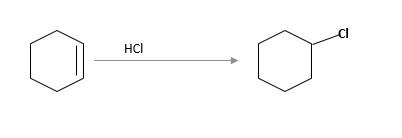
(c)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:
Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
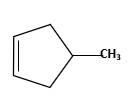 Starting alkene −
Starting alkene −
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction;
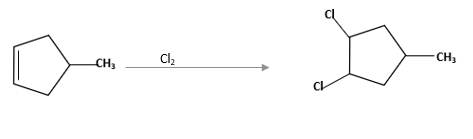
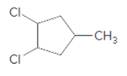
(d)
Interpretation:
The starting material and the reagents needed to prepare following alkyl halide should be determined:

Concept Introduction:
Alkenes are hydrocarbon molecules that consist a carbon-carbon double bond which has the general formula of
Reaction of alkene with
Answer to Problem 59P
Starting alkene −
Reagent -
Explanation of Solution
Unsaturated alkene molecules react with
Refer to the below reaction:
Want to see more full solutions like this?
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
Additional Science Textbook Solutions
Basic Chemistry (5th Edition)
Inorganic Chemistry
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Principles of Chemistry: A Molecular Approach (3rd Edition)
Chemistry by OpenStax (2015-05-04)
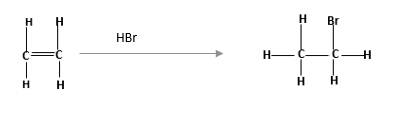
- Draw the condensed structural formula or skeletal formula, if cyclic, for the alkene that is the major product from each of the following dehydration reactions: H* а. CHз — СН2— СH-— СH2— ОН Heat ОН ОН b. H+ с. H* Heat Нeatarrow_forwardwhat is the most stable alkene?arrow_forwardName the given alkenes using systematic names. Consider alkene 1. alkene 1 name: H3C. CH2 .CH FCH2 H3C CH3 Consider alkene 2. alkene 2 name: H2 .C CH2 H2 CH3 Consider alkene 3. alkene 3 name: CH2 .CH2 H3C° IUarrow_forward
- This type of hydrocarbons contain at least one double or triple bond. Unsaturated hydrocarbons Saturated hydrocarbons Polyunsaturated hydrocarbons O Supersaturated hydrocarbons The most acidic among the hydrocarbons. Alkynes Alkenes Alkanes Arenes The number of secondary carbon(s) in this compound. CH3 CH,-C CHCH, CH3 CIarrow_forwardWhich alkane most readily undergoes thermal decomposition? Note that C-H bonds are usually stronger than C-C bonds. O ethane O dimethylpropane O propane O methylpropanearrow_forward8. Name each alkyne. A. CH3CH2CH2C=CH B. CH3CH2CH2C=CCH3arrow_forward
- Draw a Lewis Structure of an alkenearrow_forwardexplain Commercial Importance of Alkenesarrow_forwardCompounds with two carbonyl groups are named as alkane diones, for example: R 2,3-butanedione The compound above is an artificial flavor added to microwave popcorn and movie-theater popcorn to simulate the butter flavor. Interestingly, this very same compound is also known to contribute to body odor. Name the following compounds: cyclohexane-1,3-dione cycloheptane-1,4-dione gily nonane-2,8-dionearrow_forward
- What functional group distinguishes each of the following hydrocarbon derivatives? a. halohydrocarbons b. alcohols c. ethers d. aldehydes e. ketones f. carboxylic acids g. esters h. amines Give examples of each functional group. What prefix or suffix is used to name each functional group? What are the bond angles in each? Describe the bonding in each functional group. What is the difference between a primary, secondary, and tertiary alcohol? For the functional groups in ah, when is a number required to indicate the position of the functional group? Carboxylic acids are often written as RCOOH. What does COOH indicate and what does R indicate? Aldehydes are sometimes written as RCHO. What does CHO indicate?arrow_forwardName each alkyne: a.CH3CHCHCH2CH2CH3arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning  World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





