
Concept explainers
Suggest reasonable explanations for each of the following observations:
The first-order rate constant for solvolysis of
After a solution of
in aqueous sulfuric acid had been allowed to stand for
and
Treatment of
with hydrogen bromide gave the same mixture of
Treatment of
The major product in parts (c) and (d) was
Interpretation:
The reasonable explanations for each of the given observations are to be suggested.
Concept introduction:
The allyl group contains the unit
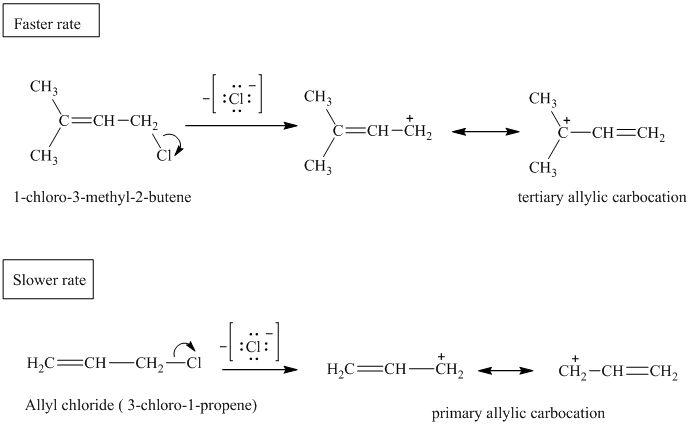
In substitution reactions, allylic halides react faster than the corresponding alkyl halides.
The primary or secondary allylic carbocations are less stable than tertiary allylic carbocations.
In allylic carbocations that are not symmetrically substituted, the two resonance structures are not equivalent and do not contribute equally to the resonance hybrid. A more stable resonance form contributes more to the resonance hybrid.
Two non-equivalent resonance forms of the allylic carbocation yield two different products.
Reactions that occur via
The regioselectivity of an
A more substituted alkene is stable and forms more readily than a less substituted alkene.
Answer to Problem 52P
Solution:
a) Resonance stabilization of the carbocation formed by the removal of chloride ion from
b) The resonance stabilization of the carbocation formed by the reaction of
c) The resonance stabilization of the carbocation formed by the reaction of
d) The resonance stabilization of the carbocation formed by the reaction of both
e)
Explanation of Solution
a) The comparison of rate constant of solvolysis of the given allylic chlorides.
The expanded structures for the two given allylic chlorides are shown below:
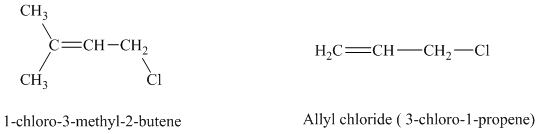
Both the allylic chlorides follow the
b) Reaction of
The expanded structure for
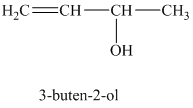
When a solution of
c) Treatment of the given compound with hydrogen bromide.
The structure for
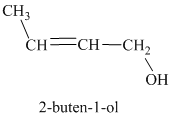
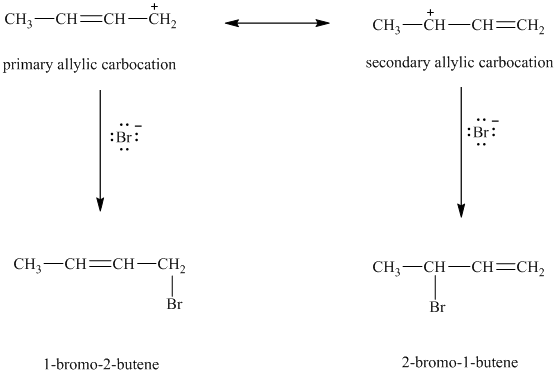
When
resonance stabilized. However, the two resonance forms for this allylic carbocation are not equivalent. Each allylic carbocation will undergo nucleophilic substitution reaction to form two different allylic bromides as shown below.
d) Treatment of
The structure for
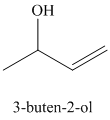
When
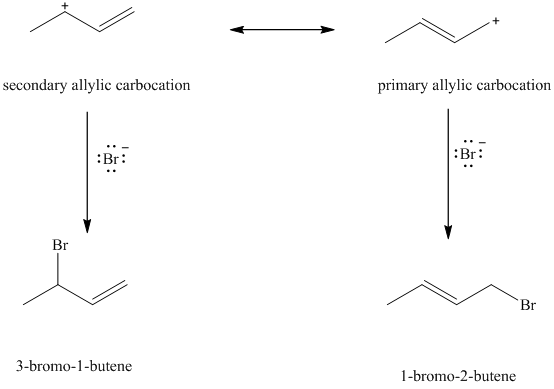
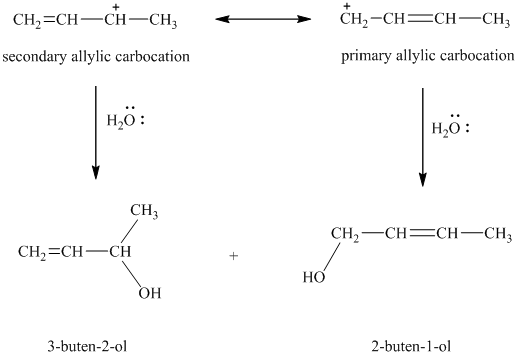
e) Major products obtained in part (c) and (d).
The products obtained in part (c) and (d) are as follows:
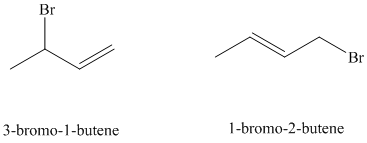
This is because this reaction follows
The major product formed in the reaction is governed by the fact that a more substituted alkene is stable and forms readily than a less substituted alkene. The bromine atom bonds to the carbon that carries the positive charge, giving
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry - Standalone book
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry - 4th edition
General, Organic, and Biological Chemistry (3rd Edition)
Organic Chemistry
Chemistry
Chemistry In Context
Chemistry: Structure and Properties (2nd Edition)
- Please solve the following reaction mechanisms for various compounds (ketone, aldehyde, alcohol, carboxylic acid, amine) in the presence of diethyl ether (solvent). These reactions occured to test their solubility in ether and all were found soluble. 0: 0 = 0 k-c-R' Ketone + CH₂ CH₂-- CH₂ CH 3 10: 11 R-C - H Aldehyde + CH2CH2-CH₂ CH 3 R- OH Alcohol + CH3 CH2 - Ộ - CH2CH3 ·0 R-C-OH Carboxylic acid + CH3CH₂-- CH₂ CH₂ H R - N-H Amine + CH CH Ộ CHỊCH, 2. ?arrow_forwardThe mechanism that allows the exothermic hydrolysis of t-butyl chloride to form t-butyl alcohol and chloride ion is as follows: (CH3)3CCl → (CH3)3C+ + Cl− (CH3)3C+ + H2O → (CH3)3COH + H+ Draw the transition state for each step.arrow_forward(a) Under certain conditions, the reaction of 0.5 M 1-bromobutane with 1.0 M sodium methoxide forms 1-methoxybutane at arate of 0.05 mol>L per second. What would be the rate if 0.1 M 1-bromobutane and 2.0 M NaOCH3 were used?arrow_forward
- CH3 CH3 Br- Br2 CH2CI2 CH3 CH3 H3C H3C Br Electrophilic addition of bromine, Br2, to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH2C12. In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to steric clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is formed. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions CH3 CH3 Br- .CH3 .CH3 H3C H3C :Br :Br:arrow_forwardWrite the reaction mechanism of the following transformations:arrow_forward(b) Write the mechanism for the formation of products in following reaction. Clearly show the intermediates in the reaction. Which product is a kinetically controlled and which one is the thermodynamically controlled product? CH3 CI. CI. A½O3, HCI H Ph-C=C-CH3 Ph CH3 Ph H.arrow_forward
- When the alkyl bromides (listed here) were subjected to hydrolysis in a mixture of ethanol and water (80% EtOH/20% H2O) at 55 °C, the rates of the reaction showed the following order: (CH3)3CBR > CH3Br > CH3CH2Br > (CH3)2CHBR Provide an explanation for this order of reactivity.arrow_forwardWhen the alkyl bromides (listed here) were subjected to hydrolysis in a mixture of ethanol and water (80% EtOH/20% H20) at 55°C, the rates of the reaction showed the following order: (CH3)3CB > CH3B > CH3CH2Br > (CH3)2CHBR Provide an explanation for this order of reactivity. Two different mechanisms are involved. (CH3)3CBr reacts by an * mechanism and apparently this reaction takes place faster. The other three alkyl halides react by * mechanism, and their reactions are slower because the 3. The reaction rates of CH3BR, CH3CH2Br, (CH3)2CHBR are affected by an * and thus their order of reactivity is CH3B > CH3CH2B > (CH3)2CHBR.arrow_forwardWrite a mechanism for the following reaction. AIBN = AIBN, heat .N. CN NC CH,SH 2.arrow_forward
- Treatment of 1,3-dichloropropane with potassium cyanide results in the formation of pentanedinitrile. The rate of this reaction is about 1000 times greater in DMSO than in ethanol. Account for this difference in rate. CI + 2 KCN NC + 2 KCI CN 1,3-Dichloropropane Pentanedinitrilearrow_forwardUseful Information: sodium metam: Commercial grade sodium metam is 33% pure sodium metam by weight and has a density of 1.2 g/mL. Hydrolysis of metam (Methyl isothiocynate) + H2S Rate constants for hydrolysis for metam (base catalyzed hydrolysis is negligible) = 300 mol -1 L. sec -1 , = 1 x 10-8 sec-1 Metam also undergoes photolysis to methyl isothiocyanate with a half-life of 1.6 hrs. Data for Sacramento River discharge Q = 75,000 L/min mean depth = 0.30 m pH = 7.8 mean width = 3.2 m dispersion coefficient D = 1.6 x 102 m2/min Assuming the spill acts acts as a single point source. How long does it take for the maximum contaminant concentration to get to Lake Shasta? If Na+ acts as conservative (i.e., it is not transformed) tracer, calculate the maximum concentration (in g/L) at Lake Shasta. (Hint: remember that sodium only constitutes a…arrow_forwardWhen 3,4-dimethyl-2-pentanol undergoes dehydration, a major alkene formed is 2,3-dimethyl-2-pentene. Give the complete mechanism for this reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
