
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.58SP
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do.
- a. CH3CH=CHCH3
- b. CH3—C = C—CH3
- c. CH2=C(CH3)2

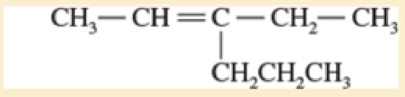
- d. CH3—CH=N—CH3
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The addition of four identical monomer molecules produces the naturally occurring molecule
shown below.
H₂C
H₂C
H₂C
CH
CH₂
CH₂
CH₂ HC
CH₂
CH₂
CH₂
CH₂
CH₂
CH
What could be the structural formula of the monomer?
A. CH2=C(CH3)CH2CH3
B. CH3CH=CHCH=CH₂
C. CH3CH₂CH₂CH=CH2
D. CH2=C(CH3)CH=CH2
CH,
Which of the following compounds can exist as cis-trans isomers?
b) For those compounds that can exist as cis and trans isomers, draw and label the isomers.
1. CH3CH=CHCH2CH2CH3
2. CH3CH2CCH2CH3=CHCH3
3. CH3CH=CHCH3
4. CH3CH2CH=CH2
Identify each pair of condensed structural formulas as
structural isomers or the same molecule.
CH3 CH CH 3
CH3 CH₂ CH₂ CH3
A.
B.
C.
CH3
CH₂ CH₂ CH3
H3C-CH₂
CH3 CH2 CH₂ - CH₂
CH3
Yvonne A
Charmita Bur
CI
Clena Khea
Dorothy
Chapter 1 Solutions
Organic Chemistry (9th Edition)
Ch. 1.2C - a. Nitrogen has relatively stable isotopes...Ch. 1.4 - Draw Lewis structures for the following compounds....Ch. 1.5 - Write Lewis structures for the following molecular...Ch. 1.5 - Circle any lone pairs (pairs of nonbonding...Ch. 1.6 - Use electronegativities to predict the direction...Ch. 1.8 - Prob. 1.6PCh. 1.9B - Draw the important resonance forms for the...Ch. 1.9B - Prob. 1.8PCh. 1.9B - Prob. 1.9PCh. 1.9B - Use resonance structures to identify the areas of...
Ch. 1.10A - Draw complete Lewis structures for the following...Ch. 1.10B - Give Lewis structures corresponding to the...Ch. 1.10B - Prob. 1.13PCh. 1.11 - Compute the empirical and molecular formulas for...Ch. 1.16 - a. Use your molecular models to make ethane, and...Ch. 1.17 - a. Predict the hybridization of the oxygen atom in...Ch. 1.17 - Predict the hybridization geometry and bond angles...Ch. 1.17 - Predict the hybridization, geometry, and bond...Ch. 1.17 - Prob. 1.19PCh. 1.17 - Allene, CH2=C=CH2, has the structure shown below...Ch. 1.17 - 1. Draw the important resonance forms for each...Ch. 1.18B - Prob. 1.22PCh. 1.18B - Two compounds with the formula CH3CH=NCH3 are...Ch. 1.19B - Prob. 1.24PCh. 1.19B - Give the relationship between the following pairs...Ch. 1 - a. Draw the resonance forms for SO2 (bonded OSO)....Ch. 1 - Name the element that corresponds to each...Ch. 1 - Prob. 1.28SPCh. 1 - For each compound, state whether its bonding is...Ch. 1 - a. Both PCl3 and PCl5 are stable compounds Draw...Ch. 1 - Draw a Lewis structure for each species a. N2H4 b....Ch. 1 - Prob. 1.32SPCh. 1 - Prob. 1.33SPCh. 1 - Draw Lewis structures for a. two compounds of...Ch. 1 - Prob. 1.35SPCh. 1 - Some of the following molecular formulas...Ch. 1 - Prob. 1.37SPCh. 1 - Give the molecular formula of each compound shown...Ch. 1 - 1. From what you remember of electronegativities,...Ch. 1 - For each of the following structures, 1. Draw a...Ch. 1 - Prob. 1.41SPCh. 1 - Prob. 1.42SPCh. 1 - Prob. 1.43SPCh. 1 - Prob. 1.44SPCh. 1 - For each pair of ions, determine which on is more...Ch. 1 - Use resonance structures to identify the areas of...Ch. 1 - Prob. 1.47SPCh. 1 - In 1934, Edward A. Doisy of Washington University...Ch. 1 - If the carbon atom in CH2Cl2 were fat. there would...Ch. 1 - Cyclopropane (C3H6, a three-membered ring) is more...Ch. 1 - Prob. 1.51SPCh. 1 - Prob. 1.52SPCh. 1 - In most amines, the nitrogen atom is sp3...Ch. 1 - Predict the hybridization and geometry of the...Ch. 1 - Draw orbital pictures of the pi bonding in the...Ch. 1 - Prob. 1.56SPCh. 1 - Prob. 1.57SPCh. 1 - Which of the following compounds show cis-trans...Ch. 1 - Give the relationships between the following pairs...Ch. 1 - Dimethyl sulfoxide (DMSO) has been used as an...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3. Which of the following compounds exists as cis-trans isomers? Draw the structures of those that do. a. CH2 = CHCl2 b. C2HSCH = CHC2H5 с. СH:CH-CHCHCHCHS d. CH;C=CCH2CH2CH3 e. CH;CH=CHC4H9 (iso) f. CH;CH2CH=C(CH3) (C2H5)arrow_forwardCH3-CH2-CH-CH3 CH3-CH-CH2-OH CH2-CH2-OH CH2-CH-OH CH3-CH2-CH2-CH2-OH CH3-CH2-O-CH2-CH3 ОН CH3 CH2.CH3 CH3 II II IV V VI I and Il are A. conformations (or conformational isomers) B. chain (skeletal) isomers C. functional isomers 31. D. positional isomers E. not isomersarrow_forwardDraw the cis and trans isomers of 2-butene, CH3CHCHCH₂. Show all hydrogen atoms. cis-2-butene Incorrect Y trans-2-butene Incorrect YA Harrow_forward
- Convert each condensed formula to a complete structure with lone pairs on heteroatoms. a. CH 3(CH 2) 8CH 3 c. CH 3CCl 3 e. (CH 3) 2CHCH 2NH 2 b. CH 3(CH 2) 4OH d. CH 3(CH 2) 4CH(CH 3) 2arrow_forwardDraw the condered structure of the ff. molecular formula a. C6H11Cl b. C4H6 c. C4H8 d. C4H9Farrow_forwardBased on the molecular formula, determine whether each compound is an alkane, alkene, or alkyne. (Assume that the hydro- carbons are noncyclical and there is no more than one multiple bond.) a. C5H12 b. C3H6 с. С-Н12 d. C11H22arrow_forward
- What is the main intermolecular forces of the following :1.) H2SO42.) N23.)KMnO44.) H2O25.)CH3CH2CH2CH3 What is the most unstable configuration form of cyclohexane?a.) chairb.) half-chairc.) boatarrow_forwarda. How many Lewis structures have the formula C4H11N? 10 b. In how many of the structures is the nitrogen atom attached to only one carbon? c. How many of them have carbon-carbon double bonds? d. How many of them have carbon-nitrogen double bonds? e. How many of them have rings? f. How many of the structures are capable of hydrogen bonding? g. How many of the structures contain a carbon atom attached to three other carbons? h. How many of the structures contain a nitrogen atom attached to three carbons? Varrow_forward1. Which of the following can exist in optically active forms. a. Cis -1, 3- Dichlorocyclohexane. b. Trans -1,3- Dichlorocyclohexane. c. Cis -1, 4- Dichlorocyclohexane. d. Trans -1,4- Dichlorocyclohexane. e. Cis -1, 2- Dichlorocyclohexane.arrow_forward
- what type of isomer? H3C CH3 H. O a. trans O b. E OC.Z O d. cisarrow_forward1. Considering compounds that have the same number of carbon atoms, explain why alkanes and cycloalkanes have different molecular formulas but alkenes and cycloalkanes have the same molecular formulas. 2. Draw the condensed or line-angle structure for an alkene with the formula C5H10. 3. Draw the condensed or line-angle structure for 4 more C5H10 isomers (2 additional alkenes and 2 cyclic isomers).arrow_forwardButane is an unbranched alkane with the condensed structure CH,CH,CH,CH,. Draw the complete structure of butane. Show all hydrogen atoms. Select Draw Rings More C H Draw the skeletal structure of butane in line-bond (line-angle) mode. Do not show hydrogen atoms. Select Draw Rings More Carrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY