
Concept explainers
a)
Interpretation:
The abbreviation of
Concept Introduction:
The seven basic units of measurement includes,
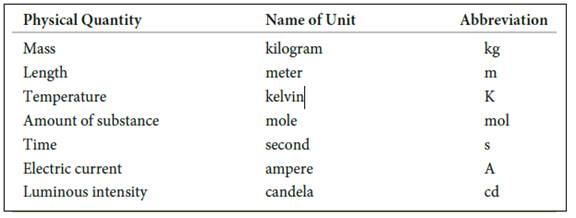
Figure 1
The symbols and their prefix of SI units are discussed below,
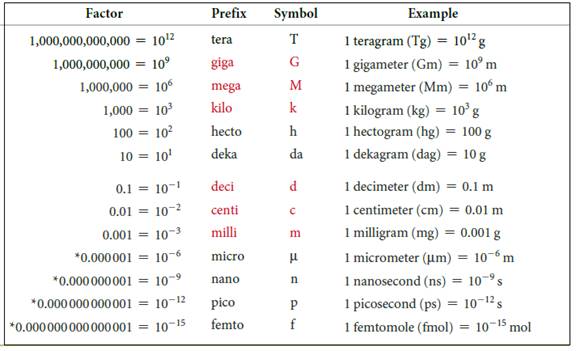
Figure 2
b)
Interpretation:
The abbreviation of
Concept Introduction:
The seven basic units of measurement includes,
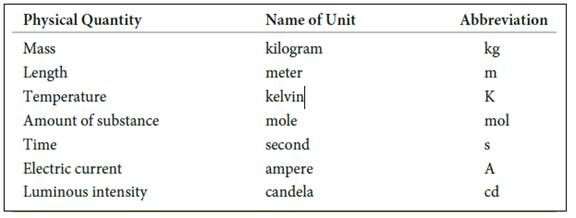
Figure 1
The symbols and their prefix of SI units are discussed below,
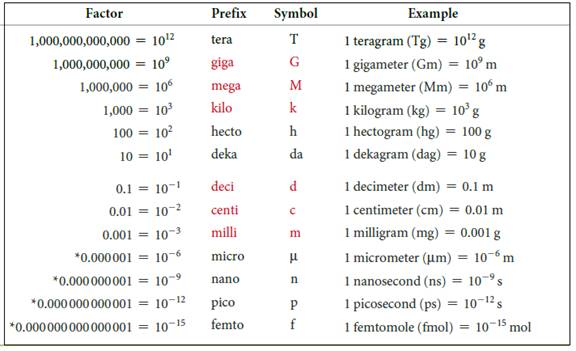
Figure 2
c)
Interpretation:
The abbreviation of
Concept Introduction:
The seven basic units of measurement includes,
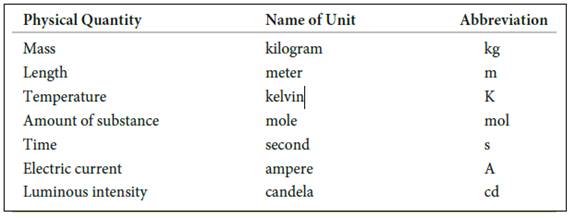
Figure 1
The symbols and their prefix of SI units are discussed below,
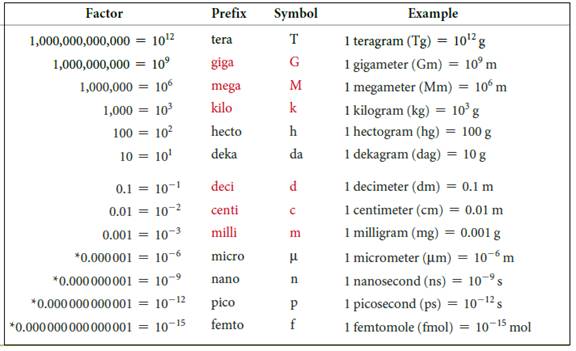
Figure 2
d)
Interpretation:
The abbreviation of
Concept Introduction:
The seven basic units of measurement includes,

Figure 1
The symbols and their prefix of SI units are discussed below,

Figure 2
e)
Interpretation:
The abbreviation of
Concept Introduction:
The seven basic units of measurement includes,
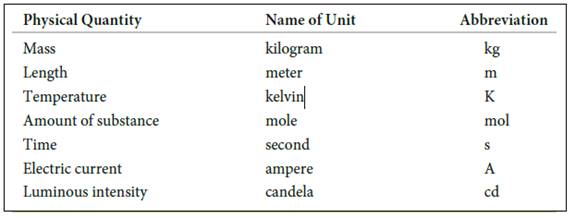
Figure 1
The symbols and their prefix of SI units are discussed below,
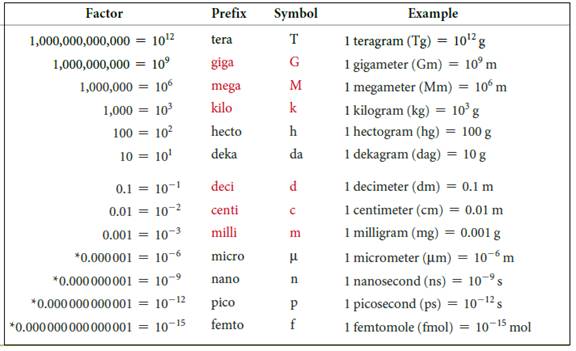
Figure 2
f)
Interpretation:
The abbreviation of
Concept Introduction:
The seven basic units of measurement includes,

Figure 1
The symbols and their prefix of SI units are discussed below,

Figure 2
Want to see the full answer?
Check out a sample textbook solution
Chapter 0 Solutions
General Chemistry: Atoms First
- What properties distinguish solids from liquids? Liquids from gases? Solids from gases?arrow_forwardWhich is larger in each pair, and by approximately how much? (a) A liter or a quart (b) A mile or a kilometer (c) A gram or an ounce(d) A centimeter or an incharrow_forwardExpress the following in scientific notation.(a) 0.0000582(b) 402(c) 7.93(d) -6593.00(e) 0.002530(f) 1.47arrow_forward
- Two beakers contain clear, colorless liquids. When the contentsof the beakers are mixed a white solid is formed. (a) Isthis an example of a chemical or a physical change? (b) Whatwould be the most convenient way to separate the newlyformed white solid from the liquid mixture—filtration, distillation,or chromatography.arrow_forwardAn astronaut on the moon places a package on a scale and finds its weight to be 19 N. (a) What would the weight of the package be on the earth? N(b) What is the mass of the package on the moon? kg(c) What is the package's mass on earth?arrow_forwardConvert the following quantities into SI units with the correct number of significant figures. (a) 5.4 in (b) 66.31 lb (c) 0.5521 gal (d) 65 mi/harrow_forward
- Q1. Which substance is a pure compound?(a) Gold (b) Water(c) Milk (d) Fruit cakearrow_forwardClassify each of the following changes as physical or chemical:(a) condensation of steam(b) burning of gasoline(c) souring of milk(d) dissolving of sugar in water(e) melting of goldarrow_forwardMusical instruments like trumpets and trombones are madefrom an alloy called brass. Brass is composed of copper andzinc atoms and appears homogeneous under an optical microscope.The approximate composition of most brass objectsis a 2:1 ratio of copper to zinc atoms, but the exact ratio variessomewhat from one piece of brass to another. (a) Would you classify brass as an element, a compound, a homogeneousmixture, or a heterogeneous mixture? (b) Would it be correctto say that brass is a solution?arrow_forward
- Perform each of the following conversions. (a) 8.43 cm to millimeters (b) 2.41 x 10 cm to meters (c) 294.5 nm to centimeters (d) 1.445 x 10* m to kilometers (e) 235.3 m to millimeters (f) 903.3 nm to micrometers (a) How many kilograms are in 1 teragram? (b) How many nanometers are in 6.50 x 10² terameters? (c) How many kilograms are in 25 femtograms? (d) How many liters are in 8.0 cubic decimeters? (e) How many microliters are in 1 milliliter? (f) How many picograms are in 1 microgram? The density of Venus is similor to that of Farth The mass of Vlenuarrow_forwardClassify each change as physical or chemical. (a) A balloon filled with hydrogen gas explodes upon contactwith a spark.(b) The liquid propane in a barbecue evaporates away becausesomeone left the valve open.(c) The liquid propane in a barbecue ignites upon contactwith a spark.(d) Copper metal turns green on exposure to air andwater.arrow_forwardFill in the blanks. (a) 700 m = km (b) 6.5 cm = mm (c) 610 mL = L (d) 3.4 L = mL (e) 550 g = kg (f) 55 kg = g (g) 28 mg = garrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

