
Concept explainers
A student names the second structure above 2,3-dimethylcyclohex-1-ene. What rule does thisviolate?
Interpretation:The rules violate by the student during naming of thegiven structure as 2, 3-dimethylcyclohex-1-ene needs to be determined.
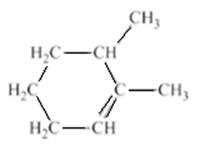
Concept introduction:
The chemical name of the compound can be defined as the name of each element or sub-compounds which are present in the chemical composition of the compound.
Answer to Problem 1CTQ
The rule of branching in the ring is violated by the student. Therefore, the name of the compound is 1, 6-dimethylcyclohexane.
Explanation of Solution
The structure of the compound is given as −
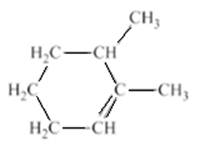
The student named the above compound as 2, 3-dimethylcyclohex-1-ene. It is to be determined that the name given by the student is correct or not. Label all the carbon atoms which are present in the ring structure to determine that the name of the compound is correct or not. The parent chain is numbered in such a way to give the lowest possible number to the double bond and these two numbers will be placed before the suffix.
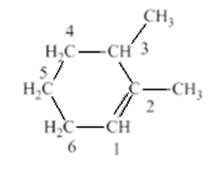
In the compound structure, the carbon with double bond should have lowest number because it has the greatest number of branches. Therefore, the numbering of the atom will start with the double bonded carbons which have methyl substituent.
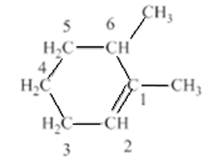
Hence, the compound will be named as 1,6-dimethylcyclohexane. In this rule of the branching in a ring containing double bond is violated by the student.
Thus, the student violated the rule of branching in the ring. Hence the name of the structure is 1,6-dimethylcyclohexane.
Want to see more full solutions like this?
Chapter NW2 Solutions
Organic Chemistry: A Guided Inquiry
- Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.arrow_forwardWe’ll see in the next chapter that there are two isomeric substances, both named 1,2-dimethylcyclohexane. Explain.arrow_forwardWhich one is more stable S2-, S4-, S6-?arrow_forward
- 1,3-dihydroxycyclohexane more stable? , is a cis isomer or trans isomer ?arrow_forwardWhat is the least and stable conformer among the choices below?arrow_forwardDraw both the condensed and line-bond structure of the following compounds: * bromocyclobutane (Br-, Bromo branch) * 5-sec-butylnonane * 4-t-butylheptane * 4-ethylcycloheptene * 4,4-dimethylpent-2-ene * 3-chloro-2,7-dimethylnon-4-yne (Cl- Chloro branch) * 1-cyclohexylbut-2-ynearrow_forward
- which drawing below represents the most stable conformation methyl cyclo hexon e of cis-1-ethyl-4. OBA #2arrow_forward4. Which of the following alkenes are stable? of ofarrow_forwardThe correct statement(s) for the following addition reactions is (are) H3C 1. BD3 THF i) M and N 2. H2O2, OH CH3 H3C CH3 1. BD3 THF ii) O and P 2. H2О, ОН- H [0] and [P] are identical molecules [M] and [N] are enantiomers First reaction yields racemic mixture; whereas second reaction yields meso isomers Total three fractions obtained, when we distilled product mixture of both the reactionsarrow_forward
- Rank the following isomeric dimethylcyclohexanes in order of decreasing stability (most stable first). T OIV > I > | > ||| O I > IV > I > III OIV > III > | > || O I > IV > III > || O III > IV> | > | A IVarrow_forwardElectron pairs CH3 .. Erase H3C - С — СІ: H3C – S- CH3 CH3 CH, CH3 THF/H,0 + H,C--CH, CH C- CH CHs CH, Use curved arrows to write the first step of this nucleophilic substitution mechanism. :ö: | :ö:arrow_forwardWhich alkene would be the LEAST stable?\arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

