
(a)
The wt % proeutectic
(a)
Answer to Problem 92AAP
The wt % proeutectic
Explanation of Solution
Refer Figure 9.43, “Aluminum-rich end of aluminum-copper phase diagram”.
Draw the plot with the notation
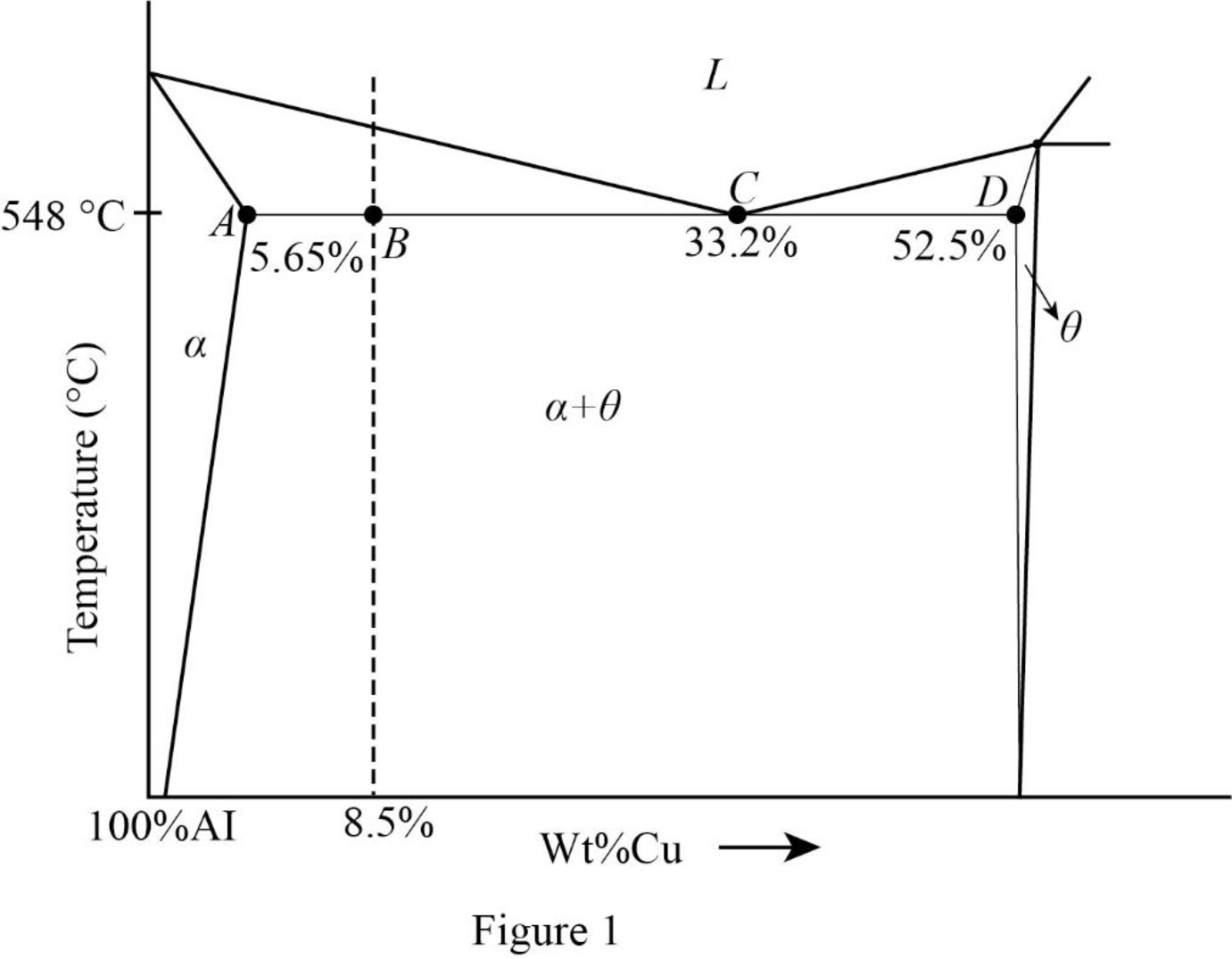
Refer Figure 1,
The weight percentage of proeutectic
Thus, the wt % proeutectic
(b)
The wt % eutectic
(b)
Answer to Problem 92AAP
The wt % eutectic
Explanation of Solution
Refer Figure 1,
The weight percentage of total
The weight percentage of eutectic
Thus, the wt % eutectic
(c)
The wt %
(c)
Answer to Problem 92AAP
The wt %
Explanation of Solution
Refer Figure 1,
The weight percentage of
Thus, the wt %
Want to see more full solutions like this?
Chapter 9 Solutions
Foundations of Materials Science and Engineering
- For a 50-50 Mg-Pb alloy, what would the percent of each phase be at 500°C, 300°C and 100°C?arrow_forward2 By using the Iron - Carbon diagram that you have. Find 5 of the following. 1- Find the Eutectic, Eutectoid temperature of cast iron with 4.3% C 2- The microstructure of Cast iron with 3.5% C. 3- Find the melting point and microstructure of steel with 0.83% C 4- Define the temperature range for Acm temperature. 5- Find the AC3, Ac¡ temperature of steel with 0.45% C 6- The Liquid and Austinite percent with chemical composition for cast iron with 2.3% C at 1300°Carrow_forwardA 45 wt% Pb-55 wt% Mg alloy is rapidly quenched to room temperature from an elevated temperature in such a way that the high- temperature microstructure is preserved. This microstructure is found to consist of the a phase and Mg2Pb, having respective mass fractions of 0.65 and 0.35. Determine the approximate temperature from which the alloy was quenched. Use Animated Figure. °℃arrow_forward
- A 10 30 70 1) For an A-B alloy of composition 25 wt% B (i) Draw the microstructure at T (just above the eutectoid temperature) and calculate the fraction of each phase in the microstructure. (ii) Draw the microstructure at T (just below the eutectoid temperature) and determine the amount of each phase, i.e., fraction of proeutectoid alpha, eutectoid alpha and eutectoid beta. 2) Provide a brief summary of phase transformations which are often observed in the iron-carbon alloy system due to various heat treatment procedures carried out to tune the microstructure. Compare mechanical strength and ductility of the materials/microstructures that form as a result of such heat treatment processes.arrow_forward3. The figure below shows the phase diagram for a hypoeutectoid plain carbon steel. Assume that point c is slightly above 723 °C and point d is slightly below 723 °C. i. ii. iii. Explain the structure at points a, b, c, and d on the diagram. Determine the wt% of proeutectoid ferrite at point c. Determine the wt% of eutectoid ferrite at point d. Temperature (°C) 1000 900 800 700 600 500 400 0 100% Fe y+ a 0.02 a + Fe, C b 0.4 723°C y + Fe, C Pearlite 0.8 Weight percent carbon Fe₂C 6.67arrow_forwardReferring to Figure 1, answer the following: What are the liquidus and solidus temperatures for an 80%Pb20%Tl alloy? What is the composition of the solid solution phase for the aforementioned alloy once we hit the liquidus temperature upon cooling? For the same alloy above, use the inverse lever rule to calculate the %solid and %liquid at 325Carrow_forward
- a. Determine the composition of each phase in a Cu-40% Ni alloy at 1300°C, 1270°C, 1250°C, and 1200°C. b. Calculate the amounts of α and L at 1250°C in the Cu-40% Ni alloyarrow_forwardiii) For a 68 wt% Zn-32 wt% Cu alloy, make schematic sketches of the microstructure that would be observed for conditions of very slow cooling at the following temperatures: 1000°C, 760°C, 600°C and 400°C. Label all phases and indicate their approximate compositions. Comportion a In 20 40 60 100 1200 |2200 H2000 Liquid 1000 - 1800 デ+ダ H1600 J400 E 1200 600 400 『+キ 600 200 400 40 60 Conpositon tet% Zroarrow_forwardA 1.5-kg specimen of a 88 wt% Pb-12 wt% Sn alloy (Animated Figure 9.8) is heated to 220°C; at this temperature it is entirely an a- phase solid solution. The alloy is to be melted to the extent that 50% of the specimen is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composition while holding the temperature constant. (a) To what temperature must the specimen be heated? i °C (b) How much tin must be added to the 1.5-kg specimen at 220°C to achieve this state? i kgarrow_forward
- A steel alloy is known to contain 93.8 wt% Fe, 6.0 wt% Ni, and 0.2 wt% C. Assume that there are no alterations in the positions of other phase boundaries with the addition of Ni. (a) What is the approximate eutectoid temperature of this alloy? i °C (b) What is the proeutectoid phase when this alloy is cooled to a temperature just below the eutectoid? (c) Compute the relative amounts of the proeutectoid phase and pearlite. Wa Wp : iarrow_forwardA binary alloy having 28 wt% Cu and balance Ag solidifies at 779°C. The solid consists of two phases a and B. Phase a has 9% Cu whereas phase ß has 8 % Ag at 779°C. At room temperature these are pure Ag and Cu respectively Sketch the phase diagram. Label all phases and temperatures. The melting points of copper and silver are 1083 and 9610 respectively s Pà) Estimate the amount of a and B in the above alloy at 779°C and at room temperature.) b) Calculate the mass fraction of phases for 40% Cu alloy at 300 °C and TR c) What is the composition of the alloys in (b)arrow_forwardBy using the Iron - Carbon diagram that you have. Find 5 of the following. 1- The microstructure of Cast iron with 3% C. 2- The Liquid and Austinite percent with chemical composition for cast iron with 2% C at 1300°C 3- Find the melting point and microstructure of steel with 0.83% C 4- Define the temperature range for Acm temperature. 5- Find the Ac3, Acl temperature and microstructure of steel with 0.4 % C 6- Find the Eutectic, Eutectoid temperature and microstructure of cast iron with 4.3% Carrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





