
Concept explainers
Interpretation: The given reaction needs to completed by adding curved arrow and drawing the product formed.
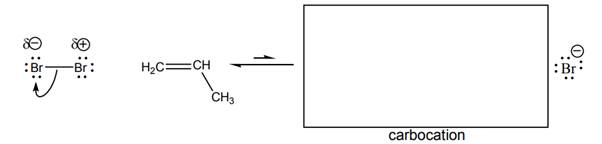
Concept Introduction: Curved arrows are used in organic reactions to show the movement of electrons. Here, electrons move from electron rich species to electron deficient species. A carbocation is formed when valency of carbon is not completed and it has a positive charge on it. The order of stability is such that tertiary carbocation is highly stable and primary carbocation is least stable.
Answer to Problem 1CTQ
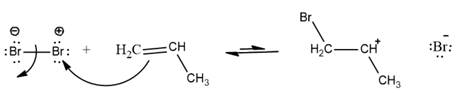
Explanation of Solution
In the given reaction, Br-Br bond breaks resulting formation of 2 Br with partial positive and partial negative charge. The bromine with partial positive charge can attack on double bond of
The movement takes place from negative charge species to positive charge. Here, double bond in alkene is considered as electron rich thus, double bond attacks on Br with partial positive charge. This results in the formation of secondary carbocation. The overall reaction can be represented as follows:
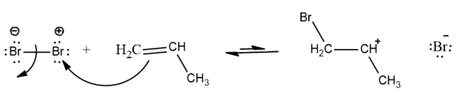
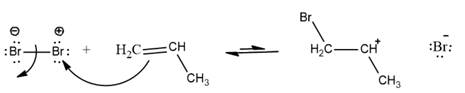
The complete reaction can be represented as follows:
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry: A Guided Inquiry
- Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HCI You do not have to consider stereochemistry. • Do not include anionic counter-ions, e.g., I, in your answer. • • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right Separate resonance structures using the symbol from the drop-down menu. ← - CHA ? n ChemDoodlearrow_forwardDraw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HBr • You do not have to consider stereochemistry. Do not include anionic counter-ions, e.g., I', in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate resonance structures using the + symbol from the drop-down menu.arrow_forwardDraw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HCI • You do not have to consider stereochemistry. • Do not include anionic counter-ions, e.g., I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate resonance structures using the → symbol from the drop-down menu. H₂ H₁₂ C C HECH H₂ C H2Cl H ? ▾ n CH₂ H₁₂ C HE HC CH H₂ H2 ChemDoodle H₁₂ CH2 HC C Cl H ? ChemDoodle F n [Farrow_forward
- [Review Topics] [References] Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. 0 / + . You do not have to consider stereochemistry. . Do not include anionic counter-ions, e.g., I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right co Separate resonance structures using the →→ symbol from the drop-down menu. 985 HCI ? Sn [Farrow_forwardDraw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HCI •You do not have to consider stereochemistry. Do not include anionic counter-ions, e.g., I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom Separate resonance structures using the → symbol from the drop-down menu. • Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HCI • You do not have to consider stereochemistry. • Do not include anionic counter-ions, e.g., I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom righ Separate resonance structures using the symbol from the drop-down menu. • - Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HBr • You do not have to consider stereochemistry. Do not include anionic counter-ions, e.g., I, in your answer. •…arrow_forwardCurved arrows, lone pairs, and formal charges are missing from the attached reactions. Add curved arrows for the forward reaction and add any needed lone pairs and formal chargesarrow_forward
- Add curved arrows to show the forming and breaking of bonds in the reaction below. C с Ċ Add/Remove steparrow_forwardCurved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the reactant and product of this hydrohalogenation reaction. Include all lone pairs.arrow_forwardDraw both resonance structures of the most stable carbocation intermediate in the reaction shown HCI - You do not have to consider stereochemistry. -Do not include anionic counter-1ons, e.g., I, in your answer. - Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. - Separate resonance structures using the symbol from the drop-down menu. y自ノ ee P. opy aste [F CHarrow_forward
- Need to check answer 1.Borane (BH3) adds to alkenes to form an alkylborane. In the first box draw the mechanism arrows, and in the second box draw the correct product. Be sure to add lone pairs of electrons and nonzero formal charges to all species.arrow_forwardDraw the structure of the alkene that will give the alcohol in Figure 10 as the main product.arrow_forwardUsing arrows, show the electron rearrangement that takes place in the reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
