
Concept explainers
(a)
Interpretation:
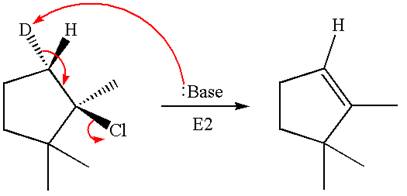
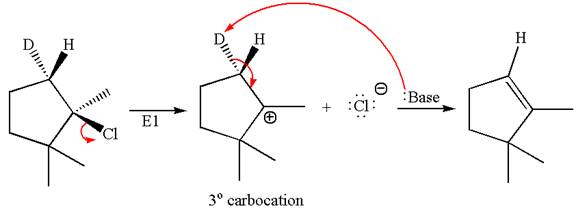
The detailed mechanism for the given reaction occurring via
Concept introduction:
The deuterium
Answer to Problem 8.64P
The
Explanation of Solution
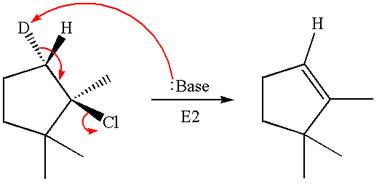
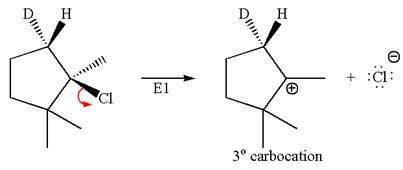
The given reaction equation is
Here, the chlorine atom is the leaving group, which can be eliminated along with either
In the given substrate, the leaving group
The product formed for the given reaction from
(b)
Interpretation:
The detailed mechanism for the given reaction occurring via
Concept introduction:
The deuterium
Answer to Problem 8.64P
The
Elimination of
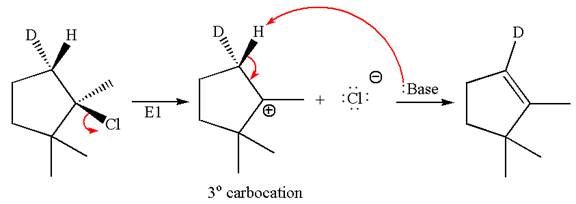
Elimination of
Explanation of Solution
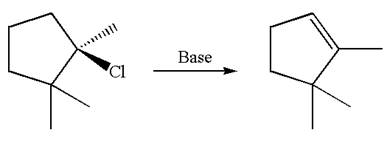
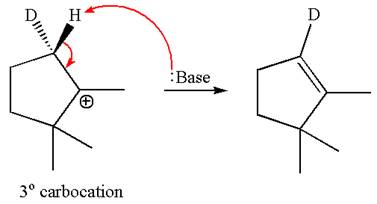
The given reaction equation is
Here, the chlorine atom is the leaving group, which can be eliminated along with either
According to
In the second step, both
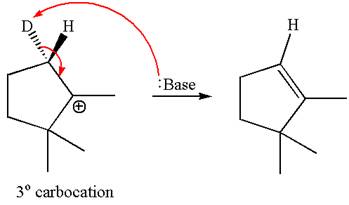
The product formed for the given reaction from
(c)
Interpretation:
The molar mass of each product from
Concept introduction:
The molar mass is the sum of the
The
Answer to Problem 8.64P
The molar masses of the products formed by
Explanation of Solution
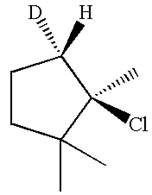
The given reaction equation is
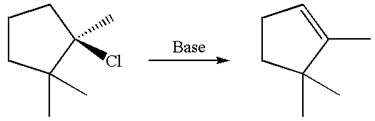
Here, the chlorine atom is the leaving group, which can be eliminated along with either
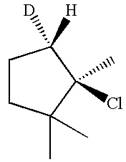
In the given substrate, the leaving group
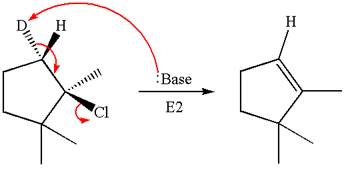
The molecular formula for this product is
According to
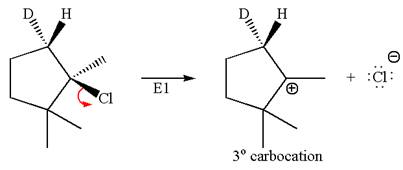
In the second step, both
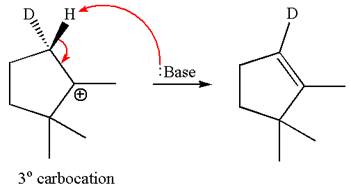
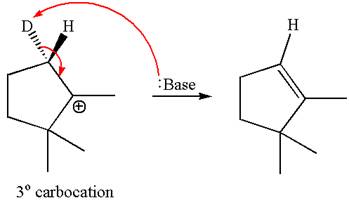
The molecular formulae for both the products formed by
Hence the molar mass of the product
The molar masses of each product formed by
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Determine whether the addition of a nucleophile to the O atom of a C=0 group is allowed or forbidden. If you determine that it is allowed, would you expect that elementary step to take place? Why or why not?arrow_forwardThe reaction shown involves an acid chloride, draw the organic product and select the inorganic species present at the end of the reaction. The structure of the acid chloride is drawn for you as a starting point, so you need to modify it to draw the reaction product. May you also draw the mechanism please to show how you got the organic product with the inorganic.arrow_forwardThe initial rates for the following elimination reaction were measured under different concentrations of the substrate and base (water); the data are tabulated at the right. Do the data suggest an E1 reaction or an E2 reaction? Trial Number [R-OCH3] [H,O] Rate (M/s) 1 0.010 M 0.45 M 9.50 × 10-4 H,O 0.020 M 0.45 M 1.85 × 10-3 OCH3 CH3CN, H 3 0.020 M О.22 М 1.85 × 10-3arrow_forward
- Each pair of reactions, 1 and 2, will both go by the same mechanism, Sn1 or S2, say which. Explain why your choice of mechanism. Also, say which of the two in each you expect to go faster, or the same, and explain why your choice goes faster.arrow_forwardWhich mechanism(s) would be favored under the following reaction conditions?arrow_forwardHow do we know whether a reaction occurs by the E1 or E2 mechanism?arrow_forward
- If Hj is eliminated from the carbocation shown here in an electrophile elimination step, then three possible constitutional isomers can form. Draw the mechanism for the formation of all three of those products. H20 +arrow_forwardWhich of the following two reactions would you predict to go faster and why?.arrow_forwardDraw the mechanism and the energy diagram for the reaction shown below. Include any resonance structures for the intermediates of the reaction. H3O+arrow_forward
- Does the reaction likely proceed by the Su1, Su2, E1, or E2 mechanism?arrow_forwardFor the given SN2 reaction, draw the organic and inorganic products of the reaction, and identify the nucleophile, substrate, and leaving group.arrow_forwardDetermine the mechanism and product for the given reaction by adding atoms, bonds, nonbonding electron pairs, and curved arrows. Acid and water are added in the second step. CH₂CH₂MgBr 2 Step 1: Add curved arrows. * MB Br → product Et₂ O Step 2: Aqueous workup. Complete the intermediate, then add curved arrows for the workup step. H : CLT: Mg Brarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
