
Concept explainers
(a)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place for this substrate.
The rearrangement can be drawn using curved arrow notation as
Explanation of Solution
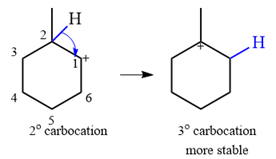
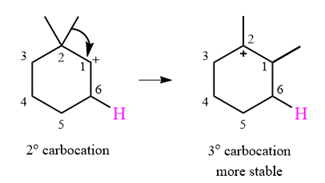
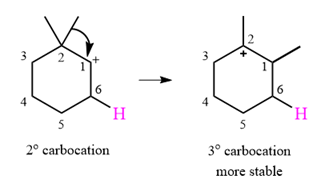
The given substrate initially forms the carbocation shown below in an
It is a relatively stable secondary carbocation. It will undergo a rearrangement only if it leads to the formation of a more stable tertiary carbocation. Two
Therefore, a carbocation rearrangement is possible in this case.
The rearrangement can be drawn using the curved arrow notation as
Formation of a more stable tertiary carbocation results in the rearrangement for this substrate.
(b)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
There will be no carbocation rearrangement in the case of this substrate.
Explanation of Solution
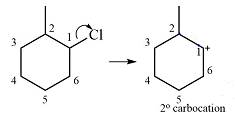
The substrate and the carbocation that it will be formed initially in an
The carbocation initially formed is a relatively stable secondary carbocation. There are two hydrogen atoms on adjacent carbons (C2 and C6) that can undergo a
Therefore, a carbocation rearrangement will not take place in this case.
There is no carbocation rearrangement for this case as there is no gain in stability.
(c)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will not take place in this case.
Explanation of Solution
The given substrate initially forms the carbocation shown below in an
The carbocation formed is a tertiary carbocation, the most stable one. Therefore, a rearrangement will occur only if it leads to another tertiary carbocation that is further stabilized by resonance. No resonance stabilization is possible here as there are no double bonds in the carbocation.
Therefore, a carbocation rearrangement will not take place in this case.
Carbocation rearrangement is not possible because the one initially formed is a tertiary carbocation.
(d)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
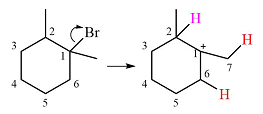
A carbocation rearrangement will take place in this case.
The curved arrow notation for the rearrangement can be drawn as
Explanation of Solution
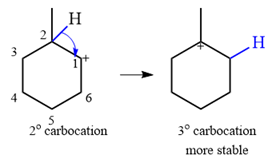
The substrate shown and the carbocation that it will initially form in this case is
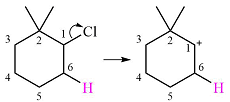
The carbocation initially formed is a relatively stable secondary carbocation. It will undergo a rearrangement only if it leads to the formation of a resonance stabilized secondary carbocation or a tertiary carbocation. Only one hydride shift is possible, but it will not occur as the carbocation formed will be a similar secondary carbocation.
A methyl shift, from C2 to C1 will, however, lead to the formation of a tertiary carbocation.
Therefore, a carbocation rearrangement will take place in this case.
The curved arrow notation for this rearrangement can be drawn as
A
(e)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
The curved arrow notation for this rearrangement is
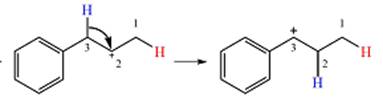
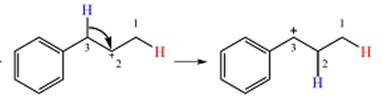
Explanation of Solution
The given substrate initially forms the carbocation shown below in an
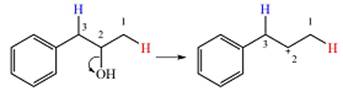
Two
The other, from C3 to C2 will result in another secondary carbocation. This would normally not lead to a more stable carbocation. However, in this case, the charge is now in a conjugated position with the
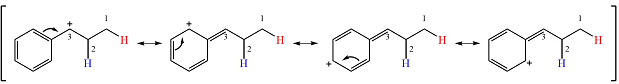
Therefore, a rearrangement will take place for this substrate.
The curved arrow notation for the rearrangement can be drawn as
Resonance can increase the stability of a carbocation as the number of atoms on which the charge is delocalized increases.
(f)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
The curved arrow notation for the rearrangement can be drawn as
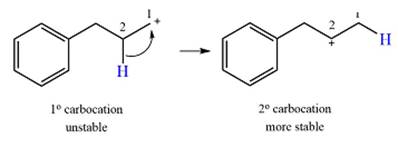
Explanation of Solution
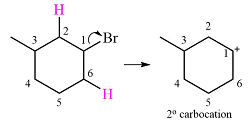
The given substrate initially forms the carbocation, as shown below, in an
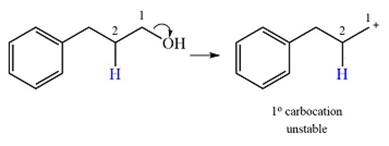
The carbocation initially formed is an unstable primary carbocation. Any rearrangement that converts it to a secondary or a tertiary carbocation will be favorable. There is only one possible
Therefore, a rearrangement of the carbocation will take place for this substrate.
The curved arrow notation for the rearrangement can be drawn as

A primary carbocation is unstable and will rearrange to a secondary carbocation where possible.
(g)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
A curved arrow notation for the rearrangement can be drawn as
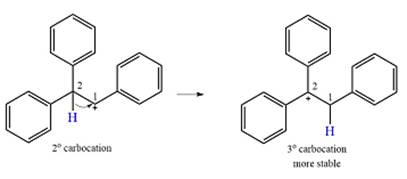
Explanation of Solution
The substrate and the carbocation that will initially be formed in an
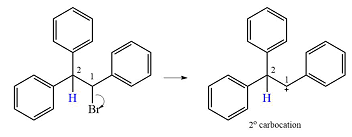
The carbocation initially formed is a relatively stable secondary carbocation. The presence of a benzene ring on the same carbon will lead to further stabilization by resonance. Resonance will delocalize the charge over a total of four carbon atoms.
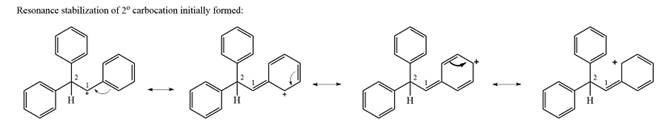
A
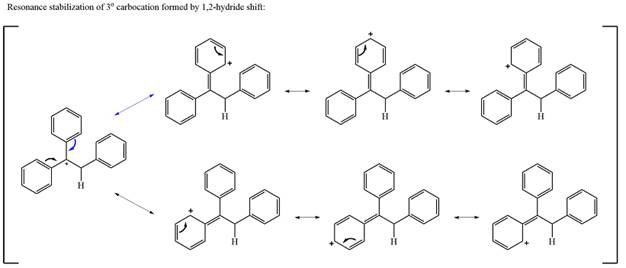
The much higher stability of this carbocation will mean a carbocation rearrangement will take place for this substrate.
The curved arrow notation for this rearrangement can be drawn as
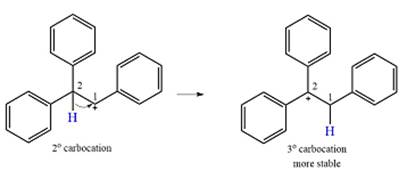
Formation of a more stable tertiary carbocation leads to the rearrangement.
(h)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
No carbocation rearrangement will take place in this case.
Explanation of Solution
The given substrate and the carbocation that it forms initially in an
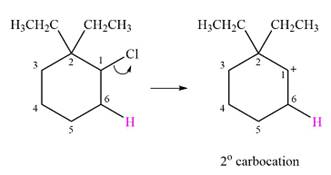
The carbocation formed is a relatively stable secondary carbocation. There is only one possible rearrangement, a
Therefore, a carbocation rearrangement will not take place in this case.
A carbocation rearrangement will not take place if it does not result in a more stable carbocation.
(i)
Interpretation:
Whether a carbocation rearrangement will take place in an
Concept introduction:
The first step in an
A carbocation may undergo rearrangement through a
Stability of carbocations increases as
Answer to Problem 8.52P
A carbocation rearrangement will take place in this case.
The curved arrow representation of this rearrangement can be drawn as
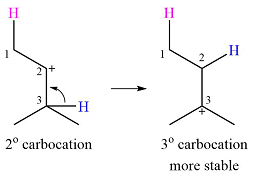
Explanation of Solution
The given substrate and the carbocation it will form in an
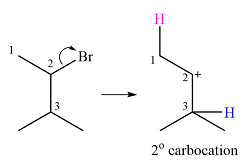
There are two possible
The second one, from C3 to C2, will result in a more stable tertiary carbocation.
Therefore, a carbocation rearrangement is possible in this case.
The curved arrow representation of this rearrangement can be drawn as

The carbocation rearrangement is possible because a more a stable carbocation is formed.
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Give two possible [1,2]-rearrangement products where an alkyl group migrates in the following compound. Motivate also which of the two products will be formed preferentiallyarrow_forward4) One of the diastereomeric cyclohexyl bromide starting materials below will readily undergo an E2 reaction while the other one will not. Fill in the requested information in boxes and lines indicated including the structure of the alkene product that does undergo an E2 reaction (use scratch paper first please). draw the expected major alkene product draw lowest E chair conformations Me. Ме Me EtO: Na Me "Ме :Br: Ме. Ме Me Me" Ме EtO: Na :Br: Required arrangement of H and LG (Br) for E2? (no acronyms). What is the rate law for and E2? On the template at left, draw all o & o* orbitals involved in the allowed E2 process.arrow_forwardConsider the molecule given below. In theory, there are only two inequivalent hydrogens in this molecule that could be substituted by Br in a free radical bromination – circle them. Put an asterisk to mark the one most likely to be substituted first. However, there are 5 possible products from free radical bromination. Draw all the products and show using arrow formalism how the intermediate radicals leading to these products formedarrow_forward
- Draw the complete, detailed mechanism (curved arrows) for the following reaction. Br 2. H₂O OKarrow_forwardThe reaction shown here is called the pinacol rearrangement. A carbocation rearrangement is believed to be involved. (a) Propose a reasonable mechanism for this reaction. (b) Suggest why the carbocation rearrangement is favorable. HO ОНarrow_forward5. Provide the product that will result for the following reaction and provide the complete mechanism. CH,N2 НО 오arrow_forward
- Consider the following reaction being performed with a low concentration. Think about what type of substitution mechanism will be favored, SN2 or SN1, and what product will result.arrow_forwardDetermine if the carbocation below is expected to undergo a rearrangement. Write NR if norearrangement will occur. If a rearrangement is expected, write the type of shift above the arrow,show the mechanism, and draw the resulting structure from the expected rearrangement.arrow_forwardChoose the compound below that gives a carbocation intermediate that undergoes rearrangement?arrow_forward
- Answer the question below the reaction. ta The reaction above proceeds through which type of mechanism? SN2 SN1 E1 E2 OH + Excess NH4C1 H₂SO4 + H₂Oarrow_forwardDraw the complete, detailed mechanism (including curved arrows) for each of the following reactions occurring via (a) an E2 mechanism and (b) an E1 mechanism. If more than one possible product can be produced from the same type of mechanism, draw the complete mechanism that leads to each one. Pay attention to stereochemistry. (i) (ii) Br NaOH NaOH ? (iii) (iv) CI KOC(CH3)3 NaOCH3 H3C CH3 (v) CH3 (vi) CI CI КОН КОН ? 'CH3 "CH3arrow_forwardDraw a complete, step-wise, curved arrow mechanism for each reaction shown below. You don't need to worry about stereochemistry for these problems. It may help if you take the following steps. 1) Find the nucleophile and the electrophile. 2) Determine the major functional group present in the nucleophile and electrophile. 3) Determine the type of reaction this particular nucleophile/electrophile pair is likely to participate in 4) Draw the mechanism that corresponds with this reaction type. a) OH cat. H2SO4 HO Cl2 b) :OHarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
