
Concept explainers
Interpretation:
The electron-poor H atom in methanol is to be identified, and the mechanism by which methanol acts as an acid in a proton transfer reaction with
Concept introduction:
An electron poor atom or a molecule is an atom or a site in a molecule that has fewer electrons than the number required for stability. In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid (proton donor) to a Bronsted–Lowry base (proton acceptor). In an elementary step, electrons tend to flow from an electron-rich site to an electron-poor site. The curved arrow notation shows the movement of valence electrons.
Answer to Problem 7.1P
The electron-poor H atom in methanol is:
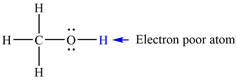
The mechanism by which methanol acts as an acid in a proton transfer reaction with
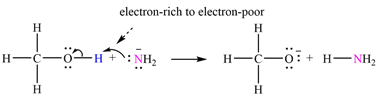
Explanation of Solution
Atoms in
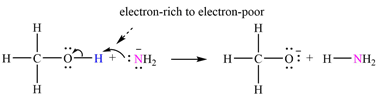
The curved arrows are drawn from the electron-rich site
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Many primary amines, RNH2, where R is a carboncontainingfragment such as CH3, CH3CH2, and so on,undergo reactions where the transition state is tetrahedral.(a) Draw a hybrid orbital picture to visualize thebonding at the nitrogen in a primary amine (just use a Catom for “R”). (b) What kind of reactant with a primaryamine can produce a tetrahedral intermediate?arrow_forwardProvide the correct reaction mechanism for the reaction in the picture.arrow_forwardProvide the reaction mechanism.arrow_forward
- Predict the products for the following reactions by showing complete reaction mechanisms. Also, identify the nucleophilic attack, loss of leaving group, proton transfer, rearrangement in the mechanism steps. Show curved arrows in the reaction mechanisms.arrow_forwardA carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.(a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyoxygen atom.(b) Compare the resonance forms with those given previously for an acid protonated on thecarbonyl oxygen atomarrow_forwardDraw the structures of hydronium ion and hydroxide ion (Show Formal Charge of atom in the structure). Show the mechanism of how they are formed using two water molecules as the reactant. (Hint: Use arrows to show the flow of electron) H2O + H2O ----> H3O+ + OH-arrow_forward
- Can i see the mechanism for this reactionarrow_forwarda)b)Convert the following c) Describe the mechanism for the following reactionarrow_forward(a) Draw the products of the proton transfer reaction shown here. (b) Draw a free energy diagram for this reaction, indicating whether it is endothermic or OH ? exothermic.arrow_forward
- I would like to understand how to mechanism of this reactions occurarrow_forwardAfter drawing a mechanism for this reaction determine which intermediate cannot be formedarrow_forwardDetermine the mechanism and product for the following reaction by adding atoms, bonds, nonbonding electron pairs, and curved arrows. Acid and water are added in the second step.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
