
Concept explainers
(a)
Interpretation:
The “
Concept Introduction:
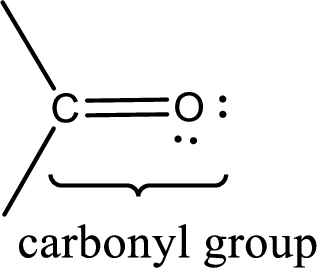
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
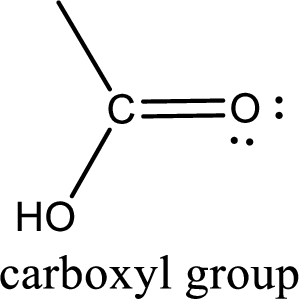
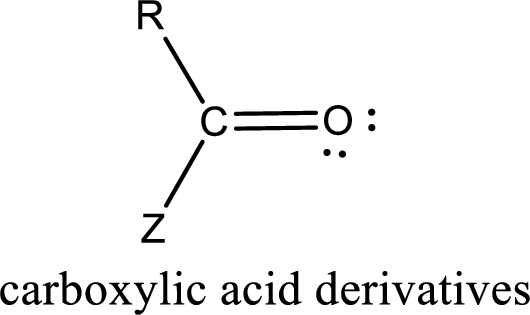
In the carboxylic acid derivatives, if the carbonyl carbon atom is bonded to a more electronegative atom means, then the bond will be polar and is it is bonded to carbon atom means then it will be nonpolar.
(a)
Answer to Problem 5.7EP
The “
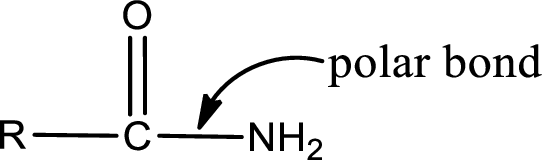
Explanation of Solution
The general structure of carboxylic acid derivatives is,
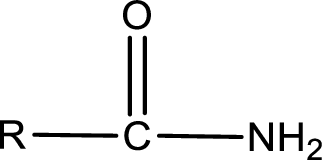
The atom in entity Z that is bonded to the carbonyl carbon atom is not a carbon atom. It is a nitrogen atom. As there is a polarity difference between carbon and nitrogen atom, the bond between carbon and nitrogen will be polar. This can be shown as given below,
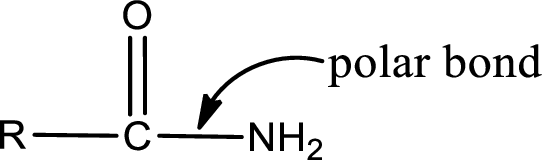
The “
(b)
Interpretation:
The “
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
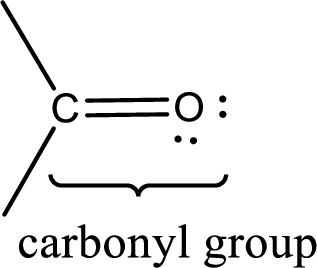
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
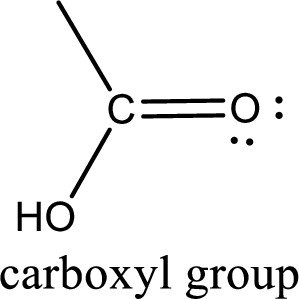
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
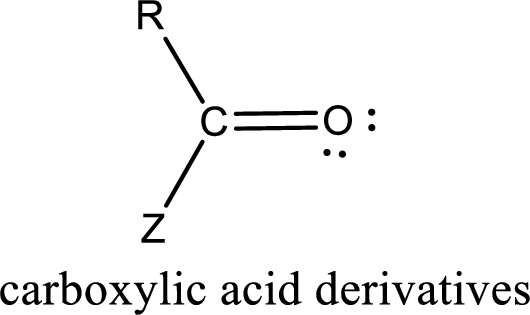
In the carboxylic acid derivatives, if the carbonyl carbon atom is bonded to a more electronegative atom means, then the bond will be polar and is it is bonded to carbon atom means then it will be nonpolar.
(b)
Answer to Problem 5.7EP
The “
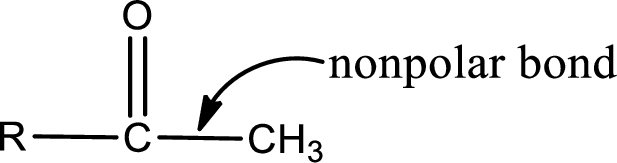
Explanation of Solution
The general structure of carboxylic acid derivatives is,
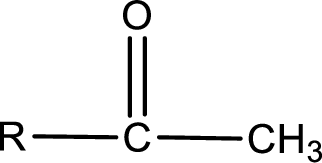
The atom in entity Z that is bonded to the carbonyl carbon atom is a carbon atom. As there is no polarity difference between carbon and carbon atom, the bond between carbon and carbon will be nonpolar. This can be shown as given below,
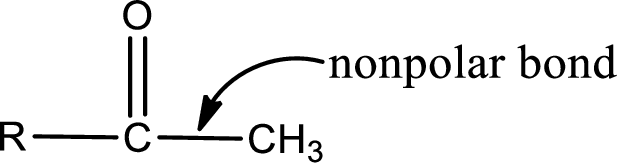
The “
(c)
Interpretation:
The “
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
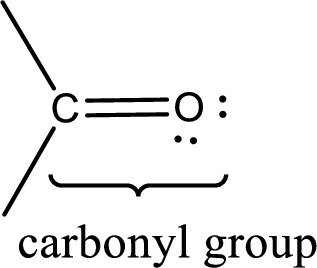
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
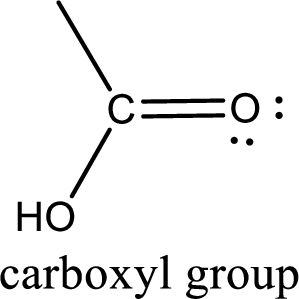
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
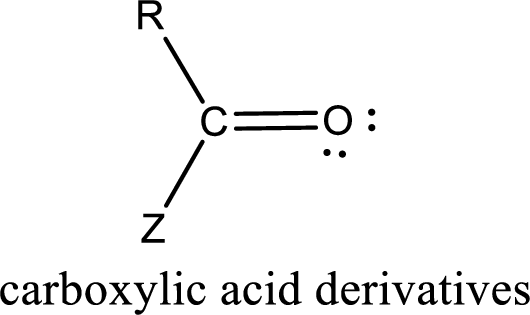
In the carboxylic acid derivatives, if the carbonyl carbon atom is bonded to a more electronegative atom means, then the bond will be polar and is it is bonded to carbon atom means then it will be nonpolar.
(c)
Answer to Problem 5.7EP
The “
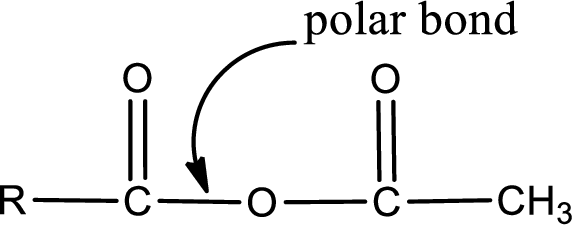
Explanation of Solution
The general structure of carboxylic acid derivatives is,
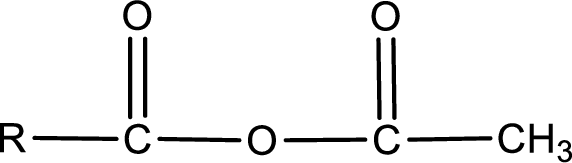
The atom in entity Z that is bonded to the carbonyl carbon atom is an oxygen atom. As there is a polarity difference between carbon and oxygen atom, the bond between carbon and oxygen will be polar. This can be shown as given below,
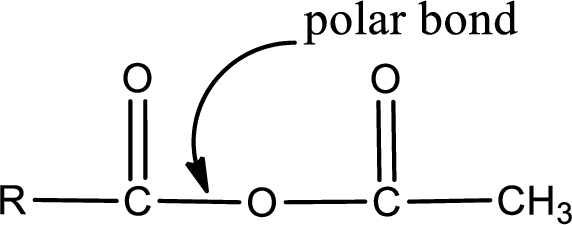
The “
(d)
Interpretation:
The “
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
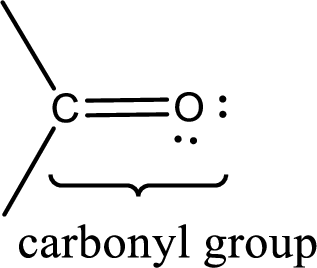
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
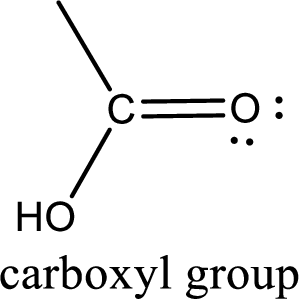
Carboxylic acid derivatives are the ones that are synthesized from or converted to a carboxylic acid. The generalized structural representation of carboxylic acid derivatives is shown below,
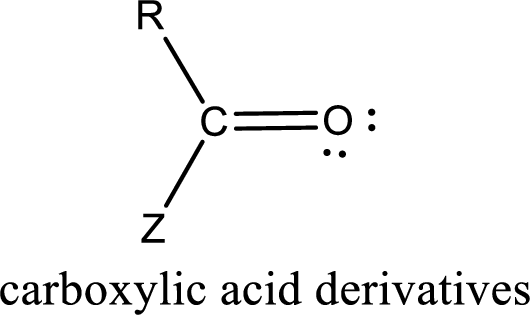
In the carboxylic acid derivatives, if the carbonyl carbon atom is bonded to a more electronegative atom means, then the bond will be polar and is it is bonded to carbon atom means then it will be nonpolar.
(d)
Answer to Problem 5.7EP
The “
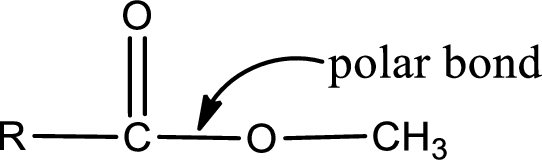
Explanation of Solution
The general structure of carboxylic acid derivatives is,
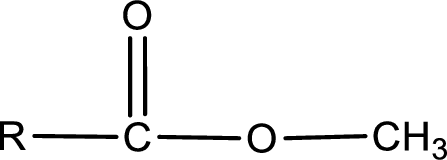
The atom in entity Z that is bonded to the carbonyl carbon atom is an oxygen atom. As there is a polarity difference between carbon and oxygen atom, the bond between carbon and oxygen will be polar. This can be shown as given below,
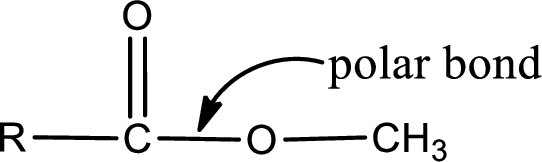
The “
Want to see more full solutions like this?
Chapter 5 Solutions
Organic And Biological Chemistry
- Fats belong to the class of organic compounds represented by the general formula, RCOOR', where R and R' represent hydrocarbon groups; therefore, fats are: a. ethers. b. soaps. c. esters. d. lipases.arrow_forwardIn terms of polarity, which carbonyl group atom possesses a a. partial positive charge b. partial negative chargearrow_forwardGive simple chemical tests to distinguish between the following pairs of compounds. isopropyl alcohol and tert-butyl alcohol methylamine and acetic acidarrow_forward
- What structural characteristic is shared by the aldehydes and the ketones? A) They both are straight chain compounds. B) Aldehydes and ketones both contain a carbonyl carbon. C) Both of these compound classes have as the smallest compound a 5 carbon skeleton. D) Aldehydes and ketones have no shared characteristics.arrow_forwardComplete each of the following reactions by supplying the missing reactant or product(s) as indicated by question marks:arrow_forwardWhich of these compounds has two chemically nonequivalent methyl groupsarrow_forward
- Draw the structural condensed formula of propanamide.arrow_forwardConsider the following two molecules and answer the following questions. Molecule A: HO- CH-CH3 CH3 Molecule B: || H,C-CH,-C-0-CH3 a) What are all of the IMFS present in molecule A? b) What are all of the IMFS present in molecule B? c) Which would have the higher boiling point, molecule A or molecule B? d) What is the relationship between molecules A & B: structural isomers, geometric isomers, identical, or not related?arrow_forwardDraw the line-angle diagram for each of the following organic compounds. For each, identify the functional group and name the class of organic compound to which it belongsarrow_forward
- Which one of the following functional groups is a base? aldehyde alcohol carboxylic acid ether aminearrow_forwardThe -COOH functional group characterizes which family of organic compounds? alcohols aldehydes amines carboxylic acidsarrow_forwardAlcohols contain which functional group? amine thiol amide hydroxylarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



