
(a)
Interpretation:
Condensed structural formula has to be drawn for the given
Concept Introduction:
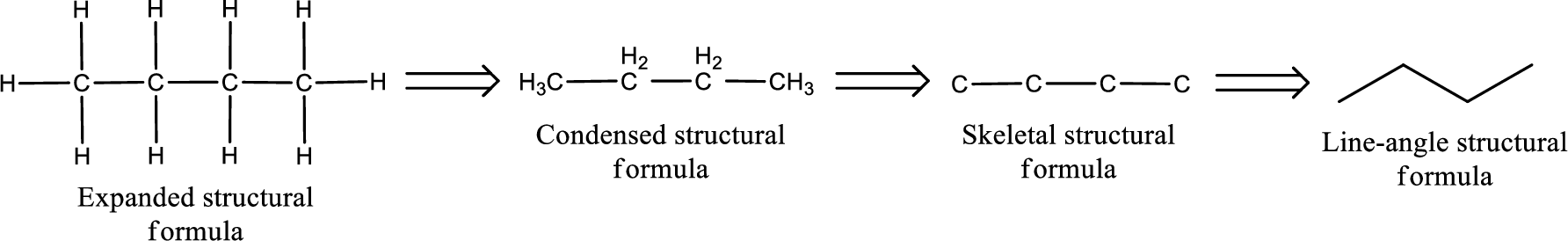
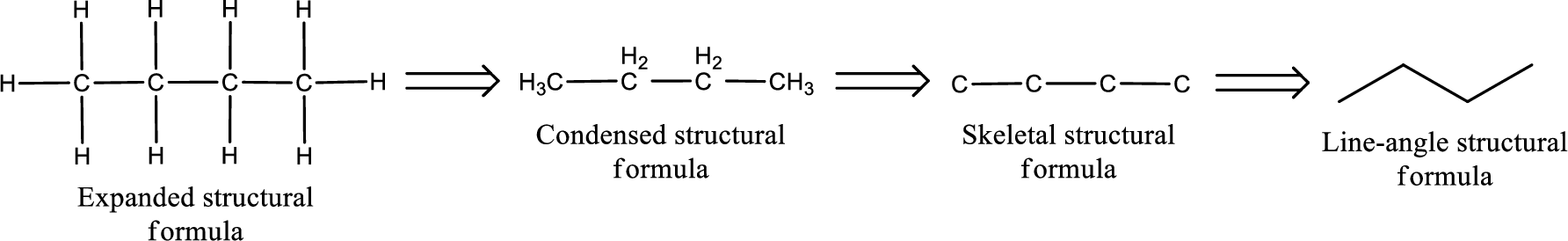
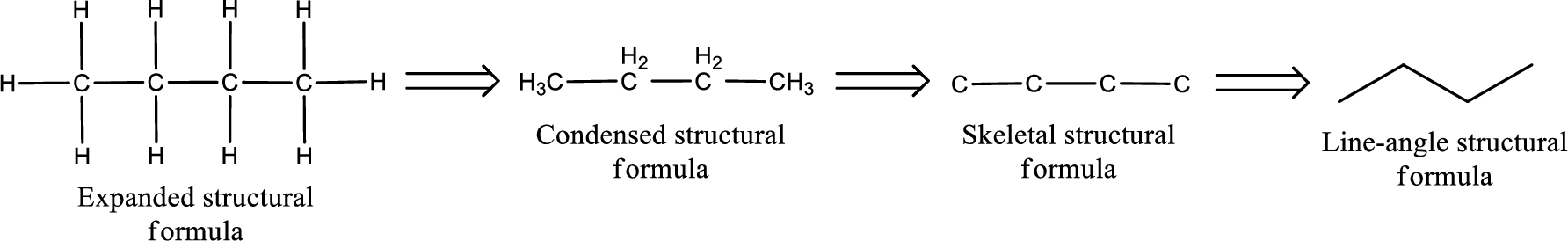
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(a)
Answer to Problem 5.30EP
The condensed structural formula is,
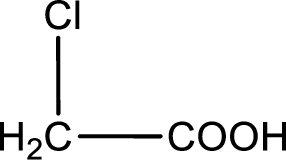
Explanation of Solution
Given description about the carboxylic acid is chloroacetic acid.
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a two carbon monocarboxylic acid. The chlorine atom is substituted in the another carbon atom. Therefore, the structure can be drawn as shown below,
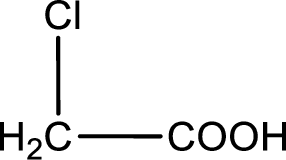
The condensed structural formula for the given carboxylic acid is drawn.
(b)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(b)
Answer to Problem 5.30EP
The condensed structural formula is,
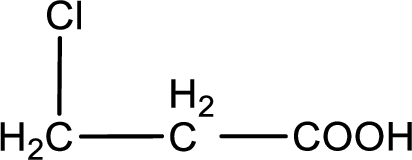
Explanation of Solution
Given description about the carboxylic acid is
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a three carbon monocarboxylic acid. The chlorine atom is substituted in the beta carbon atom. Beta carbon atom in carboxylic acid is the one that is the second carbon atom from the carboxyl group. Therefore, the structure can be drawn as shown below,
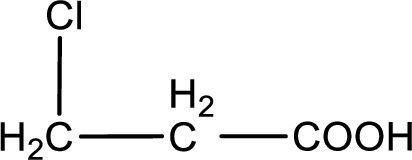
The condensed structural formula for the given carboxylic acid is drawn.
(c)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(c)
Answer to Problem 5.30EP
The condensed structural formula is,
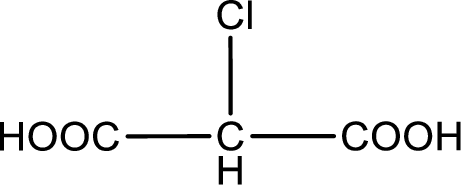
Explanation of Solution
Given description about the carboxylic acid is chloromalonic acid.
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a three carbon dicarboxylic acid. The chlorine atom is substituted in the carbon atom that is present in middle of the two carboxyl groups. Therefore, the structure can be drawn as shown below,
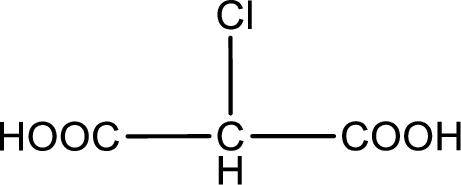
The condensed structural formula for the given carboxylic acid is drawn.
(d)
Interpretation:
Condensed structural formula has to be drawn for the given carboxylic acid.
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(d)
Answer to Problem 5.30EP
The condensed structural formula is,
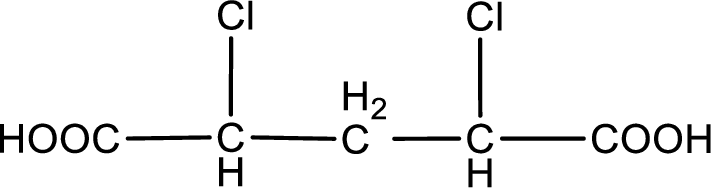
Explanation of Solution
Given description about the carboxylic acid is
In the problem statement it is given how the carboxyl group has to be denoted. From the common name it is understood that the carboxylic acid is a four carbon dicarboxylic acid. The chlorine atom is substituted in the alpha carbon atom and gamma carbon atom. Alpha carbon atom in carboxylic acid is the one that is attached to the carboxyl group and the next carbon attached to the alpha carbon atom is the beta carbon atom. The carbon atom bonded to the beta carbon atom is the gamma carbon atom. Therefore, the structure can be drawn as shown below,
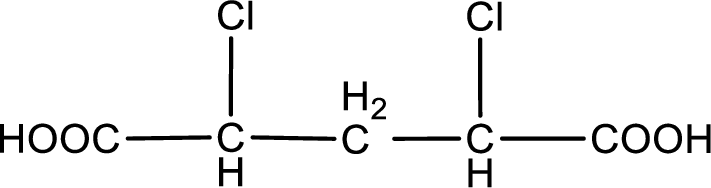
The condensed structural formula for the given carboxylic acid is drawn.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic And Biological Chemistry
- What type of chemical reaction does carboxylic acid undergo in the following situations? 1. production of carboxylate salts 2. reaction with acetyl chloride 3. one of the reactants in alcohol 4. alcohol used as reactantarrow_forwardWhat are the Generalization about Carboxylic acids and their properties.arrow_forwardAcid Alcohol Odor Structure salicylic acid methanol wintergreen ? anthranilic acid methanol Grape ?arrow_forward
- Which of the following structural formulas contains a carboxylic acid functional group? 1.CH3CH2CH2OH 2.none of these are carboxylic acids 3.CH3CH2CH2COOH 4.CH3CH2CH2COOCH3 5.CH3CH2CH2COCH3arrow_forwardthe general formula of carboxylic acidarrow_forwardgive an example of an oxidation reaction of hexanal that will result in the formation of a carboxylic acidarrow_forward
- A carboxylic acid reacts with water to form a carboxylate ion and H₂O+. Complete the reaction. reaction: CH₂COOH + H₂O= Write the IUPAC name of the carboxylate ion formed in the reaction. IUPAC name:arrow_forwardWhat organic functional group does terephthalic acid contain?arrow_forwardDefine physical properties of carboxylic acid derivatives ?arrow_forward
- Explain why carboxylic acids are weak acids.arrow_forwardHow Chemists Activate Carboxylic Acids.arrow_forward1. Which of the following structural features is possessed by aldehydes but not ketones? a. At least one hydroxyl group is bonded to the carbonyl carbon atom. b. At least one hydrogen atom is bonded to the carbonyl carbon atom. c. The carbonyl carbon atom is bonded to two other carbon atoms. d. The carbonyl carbon atom is part of a ring structure. 2. What is the IUPAC name of the following compound in the attached photo? a. 3-chloro-4-nitrobenzoic acid b. 2-chloro-4-carboxynitrobenzene c. 2-nitro-5-carboxychlorobenzene d. 2-chloro-1-nitro-4-benzoic acid 3. Which of the following is the correct bond-line structure for CH3C≡C(CH2)2CH(CH3)2? a. Structure I b. Structure II c. Structure III d. Structure IVarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





