
(a)
Interpretation:
Given compound needs to be identified whether it is monoterpene, diterpene or a sesquiterpene.
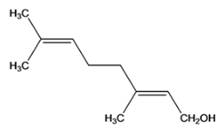
Concept Introduction :
Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(b)
Interpretation: The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
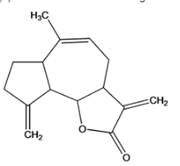 Eremanthin
Eremanthin
Concept Introduction :
Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(c)
Interpretation: The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
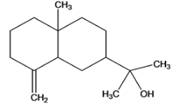
Concept Introduction : Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(d)
Interpretation:
The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
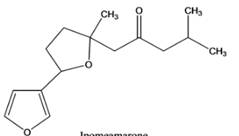
Ipomeamarone
Concept Introduction: Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(e)
Interpretation: The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
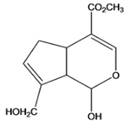
Genipin
Concept Introduction: Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(f)
Interpretation: The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
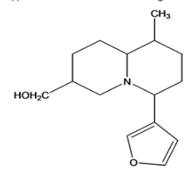
Castoramine
Concept Introduction: Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(g)
Interpretation: The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
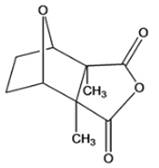
Cantharidin
Concept Introduction: Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
(h)
Interpretation: The given compound is to be identified whether it is monoterpene, diterpene or a sesquiterpene.
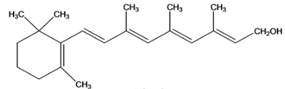
Concept Introduction: Terpenes are the natural molecules produced by plants by linking two or more molecular units of isoprene. Classification of isoprene can be given as:
| No. of carbons | No. of isoprene units | Type of terpene |
| 10 | 2 | Monoterpene |
| 15 | 3 | Sesquiterpene |
| 20 | 4 | Diterpene |
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: Structure and Function
- What is the difference in bonding and in general molecular formula between an alkene and a cycloalkane with the same number of carbon atoms?arrow_forwardWhat is the difference in bonding and in the general molecular formula between an alkene and an alkane with the same number of carbon atoms?arrow_forwardWhy are different conformations of an alkane not considered structural isomers?arrow_forward
- How does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forwardWhat is meant by the term “unsaturated hydrocarbon”? What structural feature characterizes unsaturated hydrocarbons?arrow_forwardSelect those compounds that can be correctly called unsaturated and classify each one as an alkene or an alkyne: a.CH3CH2CH3f. b.CH3CH=CHCH3g. c.h.CH2=CHCH2CH3 d.i. e.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





