
Concept explainers
(a)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.29P
The arrangement of the given compounds in decreasing order of their
Explanation of Solution
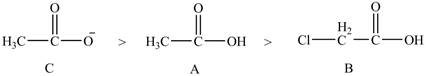
The given compounds are shown below.
Figure 1
The acidity of an atom is directly proportional to the electronegativity of an atom. The
Compound C contains
Compound A contains sulfur atom which is less electronegative than oxygen atom but it contains a chlorine atom. Chlorine atom is electronegative in nature due to which acidity of compound A increases.
Therefore, the order of acidity of the compounds is shown below.
Figure 2
The
Therefore, the order of decreasing
Figure 3
The order of decreasing
The arrangement of the given compounds in decreasing order of their
(b)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.29P
The arrangement of the order of decreasing
Explanation of Solution
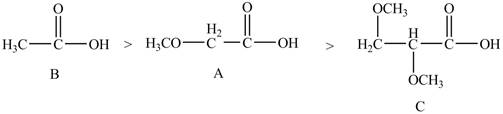
The given compounds are shown below.
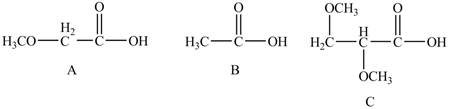
Figure 4
The acidity of an atom is directly proportional to the electronegativity of an atom. Compound C contains two
Compound A contains only one
Compound B contains
Therefore, the order of acidity of the compounds is shown below.
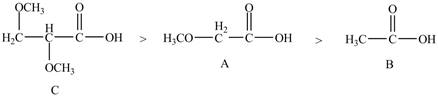
Figure 5
The
Therefore, the order of decreasing
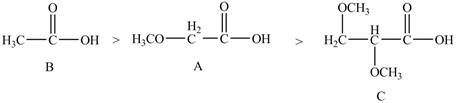
Figure 6
The order of decreasing
The arrangement of the order of decreasing
(c)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.29P
The arrangement of the order of decreasing
Explanation of Solution
The given compounds are shown below.
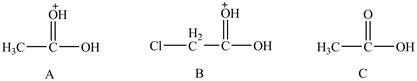
Figure 7
The conjugate bases of the compounds are shown below.
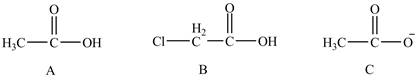
Figure 8
Compound C shown in Figure 8 is more basic than compound A and B due he presence of carboxylate ion. Compound B contains one chlorine atom which is an electron-withdrawing group. Thus, due to
The order of basicity of the compounds is shown below.
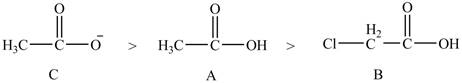
Figure 9
The order of basicity is directly proportional to the
Therefore, the order of decreasing
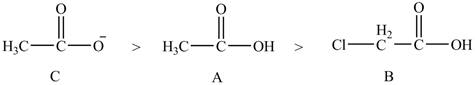
Figure 9
The order of decreasing
The arrangement of the order of decreasing
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry
- Benzoic acid (C6H5COOH) and aniline (C6H5NH2) areboth derivatives of benzene. Benzoic acid is an acid withKa = 6.3 x10-5 and aniline is a base with Ka = 4.3 x10-10. (a) What are the conjugate base of benzoic acid andthe conjugate acid of aniline? (b) Anilinium chloride(C6H5NH3Cl) is a strong electrolyte that dissociates intoanilinium ions (C6H5NH3+) and chloride ions. Which willbe more acidic, a 0.10 M solution of benzoic acid or a 0.10M solution of anilinium chloride? (c) What is the value ofthe equilibrium constant for the following equilibrium?C6H5COOH(aq) + C6H5NH2(aq) ⇌ C6H5COO-(aq) + C6H5NH3+(aq)arrow_forwardно HO но он The pK, of ascorbic acid (vitamin C) is 4.17, showing that it is slightly more acidic than acetic acid (CH3CO0H, pKa 4.74). (a) Show the fou r different conjugate bases that would be formed by deprotonation of the four different OH groups in ascorbic acid. (b) Compare the stabilities of these four conjugate bases, and predict which OH group of ascorbic acid is the most acidic. (c) Compare the most stable conjugate base of ascorbic acid with the conjugate base of acetic acid, and suggest why these two compounds have similar acidities, even though ascorbic acid lacks the carboxylic acid (COOH) group.arrow_forward(a) Find the equilibrium constant for following acid dissociation reaction at 25°C. Start your answer by writing the equation for Ka written with ΔG°. NH4+ ↔ NH3 + H+ Is your calculated equilibrium constant (Ka) consistent with the value given in the course handout? (b) Find Ka for the reaction at 10 and 40°C. Discuss how temperature affects fractionation between NH4+ and NH3. (c) NH3 is often called free ammonia as opposed to ammonium ion. While ammonia ions are relatively benign, free ammonia is known to be toxic for microbial growth at high concentration especially in high strength wastewater treatment. Briefly discuss about potential temperature effects on the toxicity in biological wastewater treatment.arrow_forward
- (b) Calculate the pH of 0.0005 mol dm-3 ethanois acid when its pKa = 4.75 CH;COOH() -> CH;COO (a H(a + Ka = pH =arrow_forwardPredict the stronger acid in each pair: (a) HNO3 or HNO2;(b) H2S or H2O; (c) H2SO4 or H2SeO4; (d) CH3COOH or CCl3COOH.arrow_forward(a) Given that Ka for acetic acid is 1.8 x 10-5 and that forhypochlorous acid is 3.0 x 10-8, which is the stronger acid?(b) Which is the stronger base, the acetate ion or the hypochloriteion? (c) Calculate Kb values for CH3COO- and ClO-.arrow_forward
- (i) Identify the acidic proton(s) for each of the following carbonyl compounds: O O ( CHỊCH, CÁCH, BỊ CHỊCHỊCH, CÁCH, (2) || (3) CHy-O-C-CH,-C-O-CH, (4) CH,-C-CH, (ii) Rank the compounds (in part (i) above) in the order of increasing or decreasing acidity by using the symbol "" in your answer.arrow_forward(a) Tsomane and Nyiko were given a task of synthesising methylenecyclohexane 2. After a brief discussion with each other, Tsomane proposed Method A to synthesise 2 from cyclohexanone 1 while Nyiko proposed Method B that started from hydroxymethylcyclohexane 3. Each student believed that their proposed method is better than the other. (Scheme below) (ii) Ph Ph Ph 640 THF A 1 B H₂SO4 100 °C 3 OH In analysing both these methods, are there other possible alkene products other than methylenecyclohexane 2?arrow_forward(a) Given that Kb for ammonia is 1.8 x10-5 and that forhydroxylamine is 1.1 x 10-8, which is the stronger base?(b) Which is the stronger acid, the ammonium ion or thehydroxylammonium ion? (c) Calculate Ka values for NH4+and H3NOH+.arrow_forward
- Acetic acid, CH3COOH, is a weak organic acid, pKa 4.76. Write an equation for the equilibrium reaction of acetic acid with each base. Which equilibria lie considerably toward the left? Which lie considerably toward the right? Q.) NaHCO3arrow_forwardWrite equations for the following acid-base reactions. Label the conjugate acids and bases, and show any inductive stabilization. Predict whether the equilibrium favors the reactants or products. Try to do this without using a table of pKa values, (a) CH3CH2OH + CH3NH- (b) F3CCOONa + Br3C¬COOH(c) CH3OH + H2SO4 (d) NaOH + H2S(e) CH3NH3+ + CH3O- (f) BrCH2CH2OH + F3C¬CH2O-(g) NaOCH2CH3 + Cl2CHCH2OH (h) H2Se + NaNH2(i) CH3CHFCOOH + FCH2CH2COO- (j) CF3CH2O- + FCH2CH2OHarrow_forwardPredict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow: (a) O2-(aq) + H2O(l)<---> (b) CH3COOH(aq) + HS-(aq)<---> (c) NO2-(aq) + H2O(l)<--->arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
