
Tutorials in Introductory Physics
1st Edition
ISBN: 9780130970695
Author: Peter S. Shaffer, Lillian C. McDermott
Publisher: Addison Wesley
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 27.2, Problem 2bTH
One mole of an ideal gas is confined to a container with a movable piston. The questions below refer to the processes shown on the PV diagram at right. Process I is a change front state X to state Y at constant pressure. Process II is a change from state W to state Z at a different constant pressure.
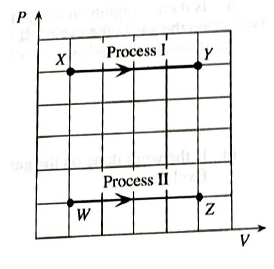
b. In the two processes, does the piston move inward, move outward, or not move? Explain.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Shown below are bar charts for processes
involving ideal gasses. For each, draw the
appropriate line or curve on the PV graph,
including an arrow for direction. Explain.
1.
Temperature Pressure Volume
bul
IF IF I F
2.
Temperature Pressure Volume
T
I FI FI F
4.
I F
IF I F
P
Temperature Pressure Volume
7 G
I FI FI F
P
3. For this, use the ideal gas law as a guide:
Temperature Pressure Volume
P
V
P
V
V
First Law of Thermodynamics
PROCEDURES: in the pictures attached
A.When gas expands, Is the (W) work done by the system or (W) work done on the system? Explain your answer.
B.Compare the temperature outside (surroundings) and inside (system) the bottle before pouring the boiling water into the bucket.
C.Compare the temperature outside (surroundings) and inside (system) the bottle after pouring the boiling water into the bucket.
Part B
A container holds a sample of ideal gas in thermal equilibrium, as shown in the
figure. (Figure 1) One end of the container is sealed with a piston whose head is
perfectly free to move, unless it is locked in place. The walls of the container readily
allow the transfer of energy via heat, unless the piston is insulated from its
surroundings.
Starting from equilibrium at point 0, what point on the pV diagram will describe the ideal gas after the following process?
"Immerse the container into a large water bath at the same temperature, and very slowly push the piston head further into the container."
• View Available Hint(s)
O point 1
O point 2
O point 3
O point 4
O point 5
O point 6
O point 7
O point 8
Submit
Previous Answers
X Incorrect; Try Again; 4 attempts remaining
The volume of the gas cannot remain constant because the piston head has moved further into the container.
Part C
Starting from equilibrium at point 0, what point on the pV diagram will describe the ideal gas…
Chapter 27 Solutions
Tutorials in Introductory Physics
Ch. 27.1 - Prob. 1aTHCh. 27.1 - In this process, which of the quantities P, V, n,...Ch. 27.1 - Consider the following incorrect student...Ch. 27.1 - Explain why it is not possible to use the ideal...Ch. 27.1 - A long pin is used to hold the piston in place as...Ch. 27.1 - A long pin is used to hold the piston in place as...Ch. 27.1 - Prob. 2cTHCh. 27.2 - Prob. 1aTHCh. 27.2 - Prob. 1bTHCh. 27.2 - Prob. 1cTH
Additional Science Textbook Solutions
Find more solutions based on key concepts
Estimate the average temperature of the air inside a hot-air balloon (see Figure 1.1). Assume that the total ma...
An Introduction to Thermal Physics
Using the method of the preceding problem, estimate the fraction of nitric oxide (NO) molecules at a temperatur...
University Physics Volume 2
Star B has an apparent magnitude of 0, which tells us how bright it appears from Earth at its true location. St...
Lecture- Tutorials for Introductory Astronomy
42. A bicycle wheel is rotating at 50 rpm when the cyclist begins to
pedal harder, giving the wheel a constant...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
30. A 3000-rn-high mountain is located on the equator. How much faster does a climber on top of the mountain mo...
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Problem 2 Suppose an irreversible process takes 1.0 mol of monoatomic ideal gas from state 1 to state 2, where pressure increases linearly with volume as shown in the diagram below. P2 Suppose that the following measurements are taken: 1 P1 Pi=101,000 N/m? Vi = 0.010 m³ p2=185,000 N/m? V2 = 0.026 m V1 V2 Calculate the following: a) work done on the system for the process. b) heat into the system for the process. c) AUsys for going from state 1 to 2. Give your answer in units of joules (J). d) ASsys for going from state 1 to 2. Give your answer in units of J/K. e) What can be known about ASsurr for this process? V (m³) p (N/m²)arrow_forward13 moles of monatomic ideal gas undergoes a thermodynamic process from point a topoint c, as shown by the PV diagram in Figure 1. i) Determine the work done ON the gas from point a to point c ii) Find the temperature of the gas at point barrow_forwardAn ideal gas undergoes two thermodynamic processes as shown in FIGURE 1. a. Name the processes AB and BC. b. If the initial temperature is 40°C, find the temperatures at B and C. c. Find the total work done d. What is the total change in internal energy for these processesarrow_forward
- In the pV diagram shown in the figure (Figure 1), 80.0 J of work was done by 0.0610 mole of ideal gas during an adiabatic process. a) How much heat entered or left this gas from a to b? Express your answer in joules. b) By how many joules did the internal energy of the gas change? Express your answer in joules. c) What is the temperature of the gas at b? Express your answer in kelvins.arrow_forward13 moles of monatomic ideal gas undergoes a thermodynamic process from point a to point c, as shown by the PV diagram in Figure 1. i) Determine the work done ON the gas from point a to point c. ii) Find the temperature of the gas at point b.arrow_forwardA gas undergoes the process shown in the diagram below. During the process AB, the internal energy of the gas decreases and a certain amount of heat Q goes out of the system for the process CA. Use this information to answer the questions below. Options for each selection is Positive, Negative, or Zero (check attached image) (a) What are the signs of W (work done by the gas), Q, and ΔU for the process CA?(b) What are the signs of W (work done by the gas), Q, and ΔU for the process AB?(c) What are the signs of W (work done by the gas), Q, and ΔU for the process BC?arrow_forward
- Referring to the figure shown, water contained in a piston–cylinder assembly, initially at 100 kPa and 25°C. The water is heated until the final temperature reaches to 320°C. Kinetic and potential energy effects are negligible.Sketch the process on T-v diagram. Determinea. The quality of the mixture when piston hits the stop.b. The final pressure.Hint: Establish the problem in three states; 1: initial, 2: when piston hits the stop, 3: at T3 = 320°C.arrow_forwardThe pV diagram in the figure (Figure 1) shows a process abc involving 0.350 mol of an ideal gas. What was the temperature of this gas at points a, b, and c? How much heat had to be put in during the process to increase the internal energy of the gas by 1.30×104 J ?arrow_forwardGive an equation for the infinitesimal change in entropy, ??, of a system in terms of the heat transfer ??? and the temperature ? at which the heat transfer occurs. Explain the sign convention.ii. A litre of water at 20 °C is placed in a fridge at 5 °C. Calculate the change in entropy of the water, including the sign, once all the water has come into thermal equilibrium.iii. Calculate the change in entropy of the fridge from part ii, including the sign.iv. Demonstrate that the Second Law has been obeyed in the process described in ii and iii.arrow_forward
- 2 moles of an ideal diatomic gas undergoes the three process cycle as depicted in the figure below. The cycle is composed of an isobaric, isovolumetric, and isothermal process. P. P. V, V. For the given parameters p.=P6=1.4 10° Pa and Va=0.7 m, V, = V.=0.35 m calculate the following. (Take the universal gas constant as R=8.314 J.mol1.K-1) a) The pressure p. = Pa b) The net work done by the gas W, J %3D net c) The heat entering the cycle QH= d) The thermal efficiency of the cycle (in percent): e = %arrow_forwardProblem 4: An ideal diatomic gas in a cylinder near room temperature undergoes the cyclic process shown. The four steps in the process are labeled A, B, C, and D. 1 (b) P2 V2 Volume Vị For each of the steps, A, B, C, D, determine whether each of the following is positive, negative, or zero: The work done on the gas. The change in energy content of the gas. The heat added to the gas. Express answers in terms of P1, P2, V1, and V2, then determine the sign. a) Make the table below showing, for each of the four steps, the sign (+, -, or 0) of W, Q and AU. AU B D b) Compute the net heat added, the net work done, and the net change in internal energy for one complete cycle.arrow_forwardA gas undergoes the cyclic process shown in the PV diagram at right. In the process abc, the system absorbs 4500 J of heat. The internal energy of the gas at a is U₁= 600 J. (b) (c) How much work is done on or by the gas from a to b? How much work is done on or by the gas when it goes from b to c ? Note that the vertical scale is measured in kilo-Pa-100 kPa -105 Pa [1 Liter = 10³ m³] 100 The net heat absorbed by the gas during the complete cycle is 1000 J. (d) Determine the heat entering or leaving in the process c to a 50 a 20 Use the First Law of Thermodynamics to determine the internal energy of the gas at c, Uc 60 C Litersarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY