
Concept explainers
a)
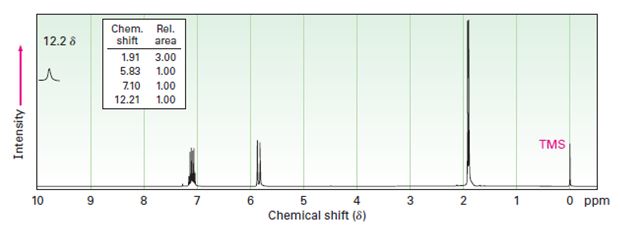
Interpretation:
Two 1HNMR spectra belonging to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) are given. Which corresponds to which is to be explained.
1HNMR: Spectrum (a): 1.91 δ (Rel.area: 3.00), 5.83 δ (Rel.area: 1.00), 7.10 δ (Rel.area: 1.00), 12.21 δ (Rel.area: 1.00).
Concept introduction:
Crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) can be easily distinguished by looking the signal for the methyl protons in these two compounds.
To explain:
Of the two 1HNMR spectra given, which belongs to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH).
b)
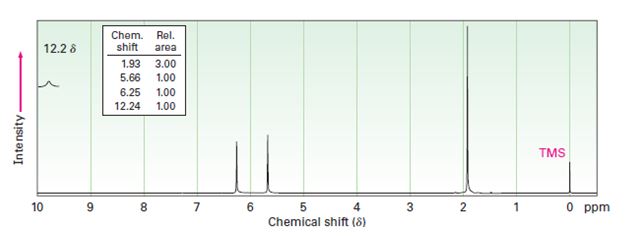
Interpretation:
Two 1HNMR spectra belonging to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) are given. Which corresponds to which is to be explained.
1HNMR: Spectrum (b): 1.93 δ (Rel.area: 3.00), 5.66 δ (Rel.area: 1.00), 6.25 δ (Rel.area: 1.00), 12.24 δ (Rel.area: 1.00).
Concept introduction:
Crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) can be easily distinguished by looking the signal for the methyl protons in these two compounds.
To explain:
Of the two 1HNMR spectra given, which belongs to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH).
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- 16-17 Propylamine (bp 48°C), ethylmethylamine (bp 37°C), and trimethylamine (bp 3°C) are constitutional isomers with the molecular formula C3HgN. Account for the fact that trimethylamine has the lowest boiling point of the three and propylamine has the highest.arrow_forwardRank the following acids in decreasing acidity. Explain your answer CH2ClCOOH, CHCl2COOH, CH3COOHarrow_forwardWhat is the correct rank of the following compounds in order of decreasing acidity? 16 NH2 II IV Multiple Choice IV < III < || < | |< || < ||| < IV III < || < | < IV I| < IV < |< |Il|arrow_forward
- What is the predicted product of the reaction sequence shown? OH excess NH3 NazCr2O7/H2SO4/H2O SOCI2 SOCI2 1. CH3CH₂MgBr 2. HO H || میں بلی HO. NH2 |||arrow_forwardUsing the data in Appendix C, determine which of the following bases is strong enough to deprotonate acetonitrile (CH3CN), so that equilibrium favors the products: (a) NaH; (b) Na2CO3; (c) NaOH; (d) NaNH2; (e) NaHCO3.arrow_forwardThe acid-catalyzed hydrolysis of an ester converts an ester into a carboxylic acid. Although there are two O atoms that can be protonated, the first step in the mechanism is believed to be protonation of the oxygen in the C=0 group. Based on charge stability, why is it favorable to protonate that oxygen? Hint: Draw out the products of each protonation. + Hо HO, НО Carboxylic acid Ester Alcoholarrow_forward
- Identify the 5-7 uses of benzoic acidarrow_forward4-Methylphenol is more acidic than ethanol (pKa 10.36 vs 16.0) , even though both contain an OH group and a methyl group. Draw the structures of the anions formed from loss of the alcoholic protons from both compounds. Use resonance to explain the difference in their respective acidities.arrow_forwardRank the following compounds in order of increasing acidity, putting the least acidic first. CH3COOH CICH₂COOH CICH₂CH₂OH I O IIIarrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

