
a)
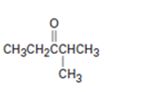
Interpretation:
The name of the ketone shown is to be given.
Concept introduction:
To give:
The name of the ketone shown.
b)
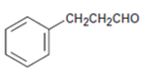
Interpretation:
The name of the aldehyde shown is to be given.
Concept introduction:
To give:
The name of the aldehyde shown.
c)
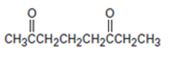
Interpretation:
The name of the diketone shown is to be given.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. The parent chain is the longest one that includes the ketone group and the numbering begins at the end nearer to the carbonyl carbon. If other functional groups are present the double bonded oxygen is considered as a substituent on the parent chain with the prefix –oxo.
To give:
The name of the diketone shown.
d)
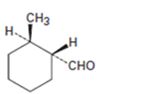
Interpretation:
The name of the aldehyde shown is to be given.
Concept introduction:
Aldehydes are named by replacing the terminal –e of the parent alkane with –al. The parent chain is the longest one that includes the -CHO group and the –CHO group is numbered as carbon 1. For cyclic aldehydes in which the –CHO group is directly attached to the ring, the suffix –carbaldehyde is used.
To give:
The name of the aldehyde shown.
e)
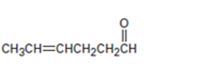
Interpretation:
The name of the aldehyde shown is to be given.
Concept introduction:
Aldehydes are named by replacing the terminal –e of the parent alkane with –al. The parent chain is the longest one that includes the –CHO group and the –CHO group is numbered as carbon 1. For cyclic aldehydes in which the –CHO group is directly attached to the ring, the suffix –carbaldehyde is used.
To give:
The name of the aldehyde shown.
f)
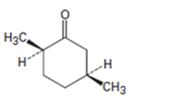
Interpretation:
The name of the ketone shown is to be given.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. For alicyclic ketones the number of carbons in the ring determines the parent name. The numbering starts from the carbonyl carbon and the numbering is done such a way that other functional groups and/or substituents get the lowest possible number.
To give:
The name of the ketone shown.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
