
Concept explainers
(a)
Interpretation:The correct name for the following sugar should be determined:
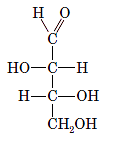
Concept Introduction: A convention used to represent a 3-D stereo formula in 2-D representation is said to be Fischer projections. In this projection, the vertical lines represent bonds below the plane of the paper and horizontal lines represents bonds above the plane of the paper.
(b)
Interpretation: The correct name for the following sugar should be determined:
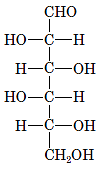
Concept Introduction: A convention used to represent a 3-D stereo formula in 2-D representation is said to be Fischer projections. In this projection, the vertical lines represent bonds below the plane of the paper and horizontal lines represents bonds above the plane of the paper.
(c)
Interpretation: The correct name for the following sugar should be determined:
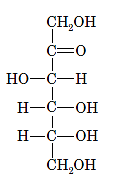
Concept Introduction: A convention used to represent a 3-D stereo formula in 2-D representation is said to be Fischer projections. In this projection, the vertical lines represent bonds below the plane of the paper and horizontal lines represents bonds above the plane of the paper.
(d)
Interpretation: The correct name for the following sugar should be determined:
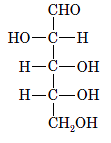
Concept Introduction: A convention used to represent a 3-D stereo formula in 2-D representation is said to be Fischer projections. In this projection, the vertical lines represent bonds below the plane of the paper and horizontal lines represents bonds above the plane of the paper.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Introduction To General, Organic, And Biochemistry
- Part 3 of 3 Modify the structure of lactulose to show the monosaccharide products that form from hydrolysis. You may draw the structures in any arrangement that you like, so long as they aren't touching. он H сн, он :☐ Он × 5 H H HO снон Сн, он H Он Н д- Н Н OH Нarrow_forward2. Sucrose is a disaccharide formed from the condensation of glucose and fructose sugars. Draw the structure of sucrose that results from the condensation reaction between the indicated -OH groups. CH2OH 1, CH,OH он 1. OH но CH,OH OH HO H он OHarrow_forward4. Complete the following Reactions thank youarrow_forward
- Consider the molecule shown below. Which statement is FALSE? H H HO H- CHO -OH -OH -H -OH CH₂OH This is an aldohexose. This is a D isomer This molecule has five chiral carbons. This is a monosaccharide. Humans can easily digest this compound.arrow_forwardWhat is the name of the sugar shown here? OH OH Он O L-xylose O D-xylose O D-erythrose.arrow_forwardHighlight each glycosidic bond in the molecule below. Then answer the questions in the table under the drawing area. 1 HO-CH, Н HO HO HO–CH, Н Н ОН Н Н Н OH Н o H ОН Н О Н Н OH CH₂ H ОН OH Н ОН HO–CH, Н Н OH H List the symbol for each type of glycosidic bond in the molecule above. For example, if there are three a,y(1-5) glycosidic bonds, write "a.r(1 - 5)". If there is more than one type of glycosidic bond, separate each symbol with a comma. Could this molecule be a tiny piece of any of the polymers listed at right? If so, check the box next to each polymer of which this molecule could be a tiny piece. O H Н ОН ОН 8 ODNA RNA O amylose glycogen O cellulose amylopectinarrow_forward
- What is the name of compound A? CHO H- OH но -H- H- OH ČH,OH A A. D-ribose B. L-talose C. D-threose D. D-xylose E. L-threose A В D Earrow_forwardName the product of the following reaction: Cl,,H*,H,Oarrow_forwardBenedict’s solution can be used to distinguish between: a) fructose and glucose b) maltose and lactose c) maltose and amylose d) amylose and amylopectinarrow_forward
- 1. Alcohol A reacts with oxygen slowly to produce a,ß-unsaturated ketone B. Applying similar reaction conditions to alcohol C, it is expected that ketone D is produced. However, D is not the product of this reaction. What is the actual product formed from the oxidation of C? Explain your reasoning. ܕ ܕܝܪ OH A B ОН 0arrow_forwardTake a look at this molecule, and then answer the questions in the table below it. CH₂OH H OH HOCH2 H I OH H H OH H OH OH H H Is this a reducing sugar? Does this molecule contain a glycosidic bond? If you said this molecule does contain a glycosidic bond, write the symbol describing it. If you said this molecule does contain a glycosidic bond, write the common names (including anomer and enantiomer labels) of the molecules that would be released if that bond were hydrolyzed. If there's more than one molecule, separate each name with a comma. yes 0 no yes no 0-0arrow_forward22 The oxidation of glucose with a weak oxidizing agent (Benedict) results in the formation of a primary alcohol on carbon #1 (CH,OH) a carboxylic acid at carbon #6 (COOH) O a bydrogenated carbon at carbon #1 (CH,OH) a carboxylic acid at carbon #1 (COOH) a carboxylic acid at both carbons #1 and #6 (COOH)arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





