
Interpretation:
The product of the reaction of cyclopentanone with the given reagents is to be predicted.
Concept Introduction:
Lithium aluminum hydride and sodium borohydride are strong reducing agents. They are inorganic compounds which are used as the reducing agents in
In the reaction of
By catalytic hydrogenation, aldehydes are reduced to primary alcohols. Grignard reagents react with ketones and aldehydes to form alcohols. These reactions are nucleophilic addition reactions. The Grignard reagent adds to the carbonyl group of aldehydes and ketones due to electronegativity difference between carbon and oxygen.
An organolithium reagent acts like a good nucleophiles and strong bases. They used for the conversion of aldehydes and ketones into primary and secondary alcohols. Acetal is an organic compound with general formula
Answer to Problem 28P
Solution:
a) The product of the reaction of cyclopentanone with lithium aluminum hydride, followed by water is shown below.
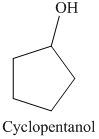
b) The product of the reaction of cyclopentanone with sodium borohydride, methanol is shown below.
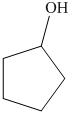
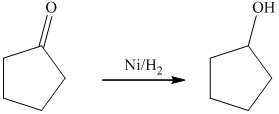
c) The product of the reaction of cyclopentanone with hydrogen (nickel catalyst) is shown below.
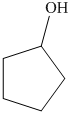
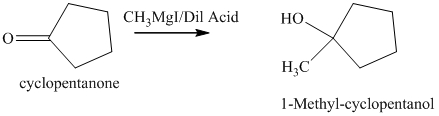
d) The product of the reaction of cyclopentanone with methylmagnesium iodide, followed by dilute acid is shown below.
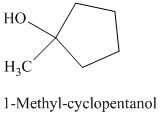
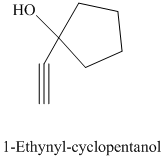
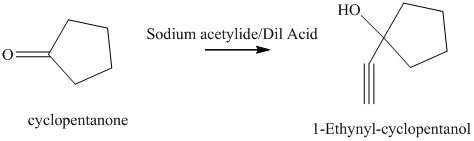
e) The product of the reaction of cyclopentanone with sodium acetylide, followed by dilute acid is shown below.
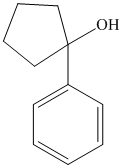
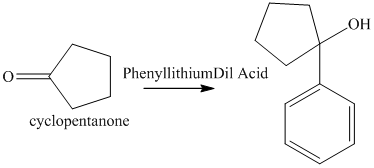
f) The product of the reaction of cyclopentanone with phenyllithium, followed by dilute acid is shown below.
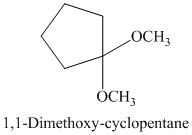
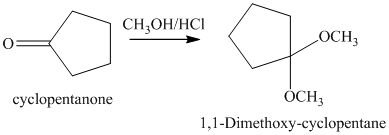
g) The product of the reaction of cyclopentanone with methanol containing dissolved hydrogen chloride is shown below.
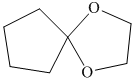
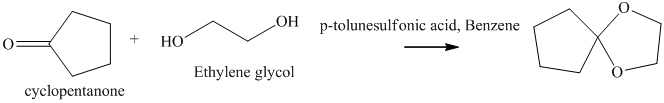
h) The product of the reaction of cyclopentanone with ethylene glycol, p-toluenesulfonic acid, benzene is shown below.
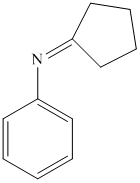
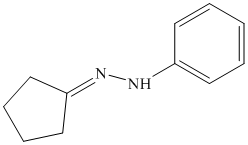
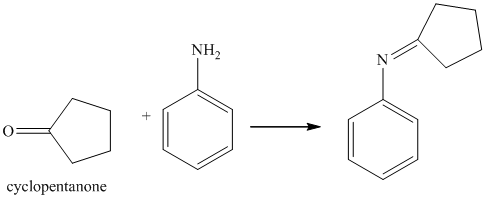
i) The product of the reaction of cyclopentanone with aniline
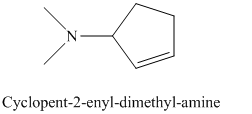
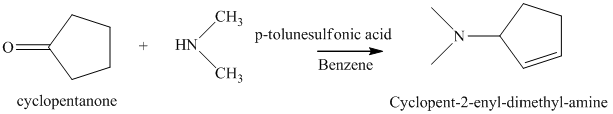
j) The product of the reaction of cyclopentanone with dimethylamine, p-toluenesulfonic acid, benzene is shown below.
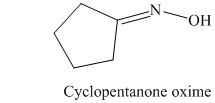
k) The product of the reaction of cyclopentanone with hydroxylamine is shown below.
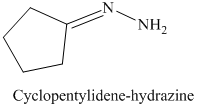
l) The product of the reaction of cyclopentanone with hydrazine is shown below.
m) The product of the reaction of cyclopentanone with product of part (l) heated in triethylene glycol with sodium hydroxide is shown below.
n) The product of the reaction of cyclopentanone with p-Nitrophenylhydrazine is shown below.
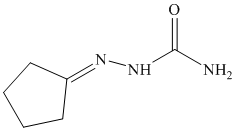
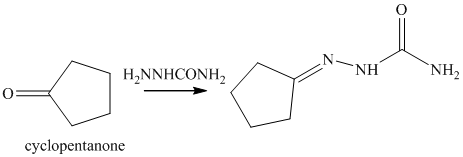
o) The product of the reaction of cyclopentanone with semicarbazide is shown below.
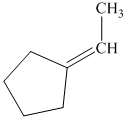
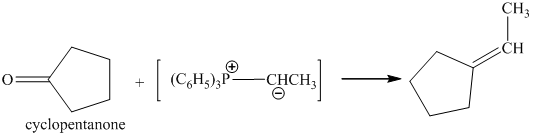
p) The product of the reaction of cyclopentanone with ethylidenetriphenylphosphorane is shown below.
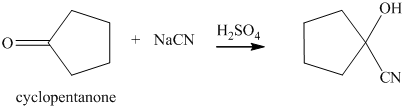
q) The product of the reaction of cyclopentanone with sodium cyanide with addition of sulfuric acid is shown below.
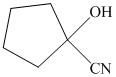
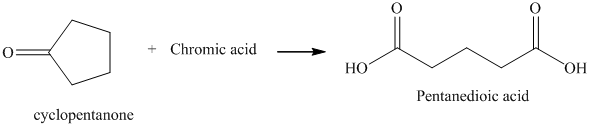
r) The product of the reaction of cyclopentanone with chromic acid is shown below.
Explanation of Solution
a) The product obtained by the reaction between cyclopentanone and lithium aluminum hydride followed by water.
The reaction of cyclopentanone with lithium aluminum hydride, followed by water gives alcohol as the final product. Lithium aluminum hydride reduces cyclopentanone to cyclopentanol. The product of this reaction is shown below.
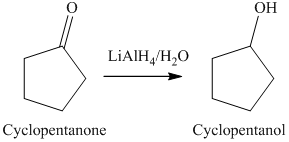
b) The product obtained by the reaction between, cyclopentanone and the reagent, sodium borohydride, methanol.
The reaction ofcyclopentanone with sodium borohydride, followed by methanol gives alcohol as the final product. The product of this reaction is shown below.
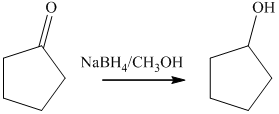
c) The product obtained by the reaction between cyclopentanone and the reagent, hydrogen (nickel catalyst).
The reaction of cyclopentanone with hydrogen in the presence of nickel catalyst gives cyclopentanol as the final product. The product of this reaction is shown below.
d) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, methylmagnesium iodide, followed by dilute acid.
The reaction of cyclopentanone with methylmagnesium iodide that is Grignard reagent, followed by dilute acid gives alcohol as the final product. The product of this reaction is shown below.
e) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, sodium acetylide, followed by dilute acid.
The reaction ofcyclopentanone with sodium acetylide, followed by dilute acid gives alcohol. The product of this reaction is shown below.
f) The product obtained by the reaction between, cyclopentanone and the reagent, phenyllithium, followed by dilute acid.
The reaction of cyclopentanone with phenyllithium, followed by dilute acid gives alcohol as the final product. The product of this reaction is shown below.
g) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, methanol containing dissolved hydrogen chloride.
The reaction of aldehydes and ketones with two equivalents of an alcohol results in the formation of acetals. The product of this reaction is shown below.
h) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, Ethylene glycol, p-toluenesulfonic acid, benzene.
In the reaction of ketone with ethylene glycol, p-toluenesulfonic acid and benzene, the protection of the carbonyl group of ketone takes place. For carbonyl protection, ethylene glycol is the commonly used group. The final product resembles like ether and known as ketal during the protection of carbonyl group using ethylene glycol. The product of this reaction is shown below.
i) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, aniline
The reaction of ketone with primary
j) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, dimethylamine, p-toluenesulfonic acid, benzene.
The reaction of ketone with secondary amine forms enamine as the final product. The reaction of cyclopentanone with dimethylamine in the presence of p-toluenesulfonic acid and benzene gives
k) The product obtained by the reaction between the given compound, cyclopentanone and the reagent, hydroxylamine.
The reaction of cyclopentanone with hydroxylamine results in the formation of cyclopentanone oxime. The product of this reaction is shown below.
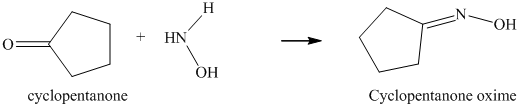
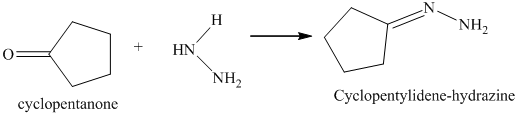
l) The product obtained by the reaction between cyclopentanone and the reagent, hydrazine.
The reaction of cyclopentanone with hydrazine gives cyclopentylidene hydrazine as the final product. The product of this reaction is shown below.
m) The product obtained by the reaction between cyclopentanone and the product of part (l) heated in triethylene glycol with sodium hydroxide.
The reaction of ketone with hydrazine gives hydrazone. The reaction of cyclopentanone with hydrazine gives cyclopentylidene hydrazine as the final product. The heating of cyclopentylidene hydrazine in triethylene glycol with sodium hydroxide forms cycloalkane as the final product. The product of this reaction is shown below.
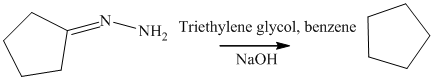
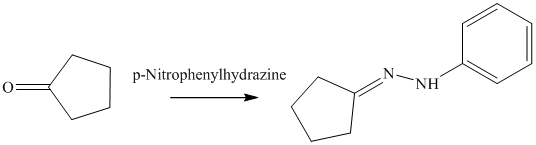
n) The product obtained by the reaction between cyclopentanone and p-nitrophenylhydrazine.
The reaction of ketone with hydrazine gives hydrazone. The reaction of cyclopentanone with p-Nitrophenylhydrazine gives hydrazone as the final product. The product of this reaction is shown below.
o) The product obtained by the reaction between cyclopentanone and semicarbazide.
The reaction of ketone with semicarbazide results in the formation of semicarbazone. The reaction of propanal with
p) The product obtained by the reaction between cyclopentanone and ethylidenetriphenylphosphorane.
The reaction of cyclopentanone with ethylidenetriphenylphosphorane gives ethylidenecyclopentane as the final product. The product of this reaction is shown below.
q) The product obtained by the reaction between cyclopentanone and sodium cyanide with addition of sulfuric acid.
The reaction of ketone with sodium cyanide results in the formation of cyanohydrin. The product of this reaction is shown below.
r) The product obtained by the reaction between cyclopentanone and chromic acid.
The reaction of ketone with chromic acid results in the formation of
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry - Standalone book
- Identify the electrophile and the nucleophile in each of the following reaction steps and then draw curved arrows to illustrate the bond-making and bondbreaking processes.arrow_forwardThe question is: "Draw the curved arrow mechanism for the reaction between pentan-2-one and (CH3)3O– in t-butanol to form an enolate. Draw all electrons and charges on both resonance structures. Then answer the question about the reaction." I got the initial arrows correct, but am not entirely sure what the carbanion intermediate would look like and then what the curved arrows would be to convert it to its final oxanion formarrow_forwardSupply the missing reagent for the following reactions.arrow_forward
- Carbon NMR Spectrum A has peaks at & 71, 40, 19, and 12. Spectrum B has peaks at 8 72, 38, 31, 29, 22, 12, and 10. Which spectrum corresponds to 1-heptanol, and which corresponds to 4-heptanol. Explain your answer using structures.arrow_forwardInclude all lone pairs and formal charges. Explicitly show any H atoms for which there is a change in bonding. Draw the mechanism, starting from the carbocation form of the acylium ion, to produce benzophenone.arrow_forwardSynthesize the following compound from cyclohexanone and any other organic and inorganic reagents you need.arrow_forward
- Consider the reaction of the cyclopentanone derivative shown below.arrow_forwardBiaryls, compounds containing two aromatic rings joined by a C–C bond, can often be efficiently made by two different Suzuki couplings; that is, either aromatic ring can be used to form the organoborane needed for coupling. In some cases, however, only one route is possible. With this in mind, synthesize each of the following biaryls using benzene as the starting material for each aromatic ring. When more than one route is possible, draw both of them. You may use any required organic or inorganic reagents.arrow_forwardA reaction similar to the one described here occurs when benzophenone is treated with the metal magnesium (pinacol reduction). Compare the mechanism of this reaction with the photoreduction mechanism. What are the differences?arrow_forward
- Here in Chapter 19, we learned that converting a ketone or aldehyde to an acetal is a good way to protect the carbonyl group, because an acetal is composed of ether linkages. In Problem 17.57, we saw that a ketone or aldehyde can be converted into an epoxide, which is a cyclic ether. Why would an epoxide be a poor choice as a protecting group?arrow_forwardProvide a mechanism for the following transformationarrow_forwardExplain why the following cyclohexanol A derivative undergoes E2 more readily than cyclohexanol B. State three reasons.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





