
Concept explainers
(a)
Interpretation:
The electron-pair geometry for the molecules,
Concept introduction:
The electron pairs in Lewis diagrams repel each other in real molecule and thus, they distribute themselves in positions around the central atoms that are as far away from one another. This arrangement of electron pairs is called electron-pair geometry. The electron pairs may be shared in covalent bond, or they may be lone pairs.
Answer to Problem 20E
The Lewis diagrams for
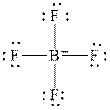 and
and 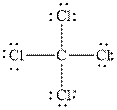
The wedge-and-dash diagrams for
 and
and 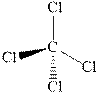
The electron pair geometry for both molecules is tetrahedral.
Explanation of Solution
To write the Lewis diagram for a compound first the number of valence electrons is to be calculated. In the molecule,
Similarly, in the molecule,
The atom which is least electronegative is the central atom. In

Figure 1
In

Figure 2
The electron-pair geometry depends on the number of electron pairs around the central atoms. In both the molecules
The wedge-and-dash diagram for the molecules

Figure 3
The wedge-and-dash diagram for the molecules

Figure 4
The Lewis and wedge-and-dash diagrams for
(b)
Interpretation:
The molecular geometry predicted by the valence shell electron-pair repulsion theory for the molecules
Concept introduction:
Molecular geometry is the precise term that is used to describe the shape of molecules and arrangement of atoms around the central atom. The molecular geometry of a molecule is predicted by valence shell electron-pair repulsion theory or in short VSEPR theory. VSEPR theory applies to substances in which a second period element is bonded to two, three, four, or other atoms.
Answer to Problem 20E
The Lewis diagrams for
 and
and 
The wedge-and-dash diagrams for
 and
and 
The molecular geometry for both molecules is tetrahedral.
Explanation of Solution
To write the Lewis diagram for a compound first the number of valence electrons is to be calculated. In the molecule,
Similarly, in the molecule,
The atom which is least electronegative is the central atom. In

Figure 1
In

Figure 2
The molecular geometry depends on the number of electron pairs as well as number of unpaired electron on the central atoms. In both the molecules
The wedge-and-dash diagram for the molecules

Figure 3
The wedge-and-dash diagram for the molecules

Figure 4
The Lewis and wedge-and-dash diagrams for
Want to see more full solutions like this?
Chapter 13 Solutions
Introductory Chemistry: An Active Learning Approach
- QUESTION 4 Give the molecular geometry and number of electron groups for BrF5. square planar, 6 electron groups T-shaped, 5 electron groups seesaw, 5 electron groups octahedral, 6 electron groups O square pyramidal, 6 electron groups QUESTION 5 Place the following in order of decreasing X-A-X bond angle, where A represents the central atom and X represents the in each molecule. CS₂ CF4 O CS2 > CF4> SC12 CF4 > SC12 > CS2 CF4> CS2 > SC1₂ ⒸCS₂ = SC12 > CF4 SC12 > CF4 > CS₂ QUESTION 6 How many of the following molecules are polar? KrBr2 SC³6 OCCl2 PBr5 2 00000 0 01 SC1₂arrow_forwardWrite the Lewis structure for the following molecule. State the electronic structure (shape based on electron pairs or bonds) AND the geometric structure (shape based on the atoms only). Include all valence electrons in your structure. State how many electrons are used to form covalent bonds in the molecule. Is the molecule polar? O2arrow_forwardUse Lewis theory to determine the formula for the compound that forms between each of the following pairs of elements. Ca and I Express your answer as a chemical formula. Na and Se Express your answer as a chemical formula. Al and O Express your answer as a chemical formula.arrow_forward
- NH3 vs BH3 NH3 BH3 polar nonpolar polar nonpolar Molecular Geometry Molecular Geometry Question 1b: How does the molecular geometry (trigonal pyramidal vs trigonal planar) affect the polarity?arrow_forwardAnswer the questions in the table below about the shape of the phosgene (COCI,) molecule. How many electron groups are around the central carbon atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central carbon atom? (You may need to use the scrollbar to see all the choices.) (choose one)arrow_forwardAnswer the questions in the table below about the shape of the sulfur hexabromide (SBr) molecule. How many electron groups are around the central sulfur atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central sulfur atom? (You may need to use the scrollbar to see all the choices.) (choose one) X Ś Um 0 Garrow_forward
- Molecule or lon Bil4 #valence BrF3 #valence BrF4 #valence e Lewis Structure Number of Regions of Electron Density on Central Atom Lone Pairs (LP) Bond Pairs (BP) Sketch possible Molecule/lon Show lone pair(s) most stable postistion Molecular Geometry (write the name)arrow_forwardDraw a Lewis structure for the compound whose skeletal structure is provided to you below. Don't forget to draw in the H atoms! CH₂ HC CH 11 HC — CH, - Draw the Lewis structure by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forwardConvert the following Lewis structure for the nitrate ion into a line structure that includes the lone pairs. Why does the nitrate ion have a -1 charge?arrow_forward
- Draw a Lewis structure for the following ions/molecules, including any resonance structures and/or formal charges. Place a box around any contributing resonance structures XeO4^-2, BrF5, CHF2CL What is the electron group geometry around the central atom for each of the above ions/molecules? For the above ions/molecules, what is the shape of the entire molecule/ion of the entire ion/molecule? Identify polar bonds with dipole arrows Indicate whether each of the ions/molecules is POLAR or NON-POLARarrow_forwardXenon can be the central atom of a molecule by expanding beyond an octet of electrons. Draw Lewis structures for the given xenon compounds. Show all lone pairs. XeF, XeF,0arrow_forwardAnswer the questions in the table below about the shape of the chlorotetrafluoride (CIF) . cation. How many electron groups are around the central chlorine atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central chlorine atom? (You may need to use the scrollbar to see all the choices.) 0 (choose one) X Sarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





