
Concept explainers
Write structural formulas and give the IUPAC names for all the isomers of
Interpretation:
The structural formulas and IUPAC names for all the isomers of
Concept introduction:
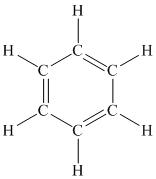
A benzene ring is a cyclic six membered ring containing single and double bonds alternately between carbon atoms. One hydrogen atom is attached to each carbon atom.
In a mono substituted benzene ring, one hydrogen atom is replaced by a substituent.
The substituent should be such that each carbon atom must have formed four bonds and other atoms, if any, should have complete octets.
Answer to Problem 33P
Solution:
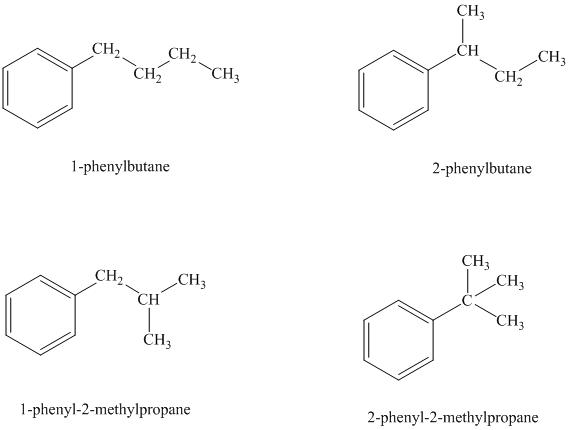
Explanation of Solution
The structure of a benzene ring is as follows:
One hydrogen atom is replaced by the group
There are four possible ways in which this group can be attached to the ring.
The structures and IUPAC names of the isomers are shown below:
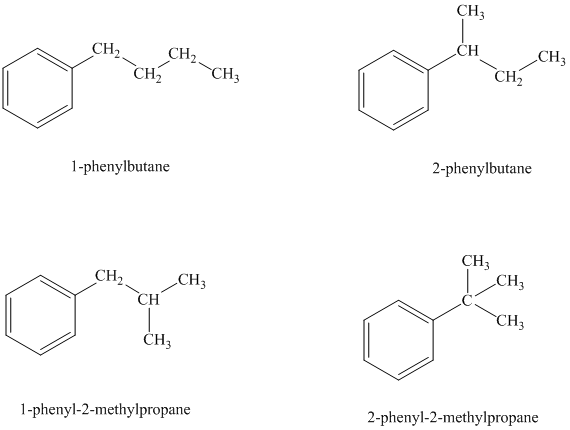
In a mono substituted benzene ring, one hydrogen atom is substituted by a substituent. An alkyl substituent can be arranged such that each carbon atom must have formed four bonds and other atoms if any should have complete octets.
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry - Standalone book
- What is the difference in bonding and in general molecular formula between an alkene and a cycloalkane with the same number of carbon atoms?arrow_forwardGive the structure of the following compounds and write their IUPAC names: Ethyl propyl ether Methyl n-butyl ether Ethyl isopropyl ether Dipropyl etherarrow_forwardStructural formula of 2-ethyl-3-methylheptanolarrow_forward
- Write structural formulas for the following compounds. 2-ethoxyoctanearrow_forwardWhat is the structure of a compound with the formula C7H14O that has an ether and cyclobutane ring, including its IUPAC name? In addition, what is the structure of the compound with the formula C7H14O that does NOT contain the functional groups: epoxide, ether, cycloalkane, alcohol, alkene. Indicate the functional groups used that are present in the structure made for this compound.arrow_forwardGive the reagent or chemical test that would differentiate the following pairs of compounds. Provide only the reagents or chemical tests discussed in the Write chemical equations for the reactions involved. toluene and benzene 1-pentyne and 2-pentyne 1-methylcyclohexane and 1-methylcyclohexenearrow_forward
- Write a condensed structural formula for a cyclic ketone with the formula C5H8Oarrow_forwardGive the reagent and the reaction conditions that would distinguish between the following compounds. Explain the principles behind the reactions. Write the chemical equation for the reactions involved. 1-chloro-3-methylbutane and 2-cyclopentenolarrow_forwardGive the structural formula (condensed or skeletal) of the given IUPAC names of saturated hydrocarbons. 1) 2-methyl-3-oxohept-4-enoic acid 2) 4-chloro-2-oxohexanoyl chloride 3) 4-nitro-4-propoxybut-1-ene 4) 1-hydroxyhex-5-yn-2-one 5) 1-methylpropyl 3-amino-3-(1-methylpropoxy)propanoate 6) 1-ethoxy-3-mercaptohexane-2-thionearrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



