
Concept explainers
Answer the following questions about aldosterone, a compound that helps to control the absorption of Na+and Cl- ions in the kidneys and, as a result, affects water retention.
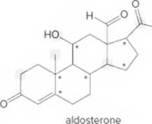
- Identify the
functional groups. - Draw in all lone pairs on O atoms.
- How many C’s does aldosterone contain?
- How many H’s are present at each C labeled with an asterisk (*)?
- Give the shape around each atom labeled in gray.
- Label all polar bonds.
(a)
Interpretation:
The functional group in aldosterone should be midentified.
Concept Introduction:
Functional groups are the atoms or group of atoms that gives chemical properties to an organic compound and also chemical reactivity centre. Examples of functional groups are aldehyde, ketone, alcohol, double bond.
Answer to Problem 83P
In aldosterone, functional groups present are alcohol, aldehyde, ketone and alkene.
Explanation of Solution
Functional groups are the atoms or group of atoms that gives chemical properties to an organic compound and also chemical reactivity centre. Structures of different functional groups are as follows:
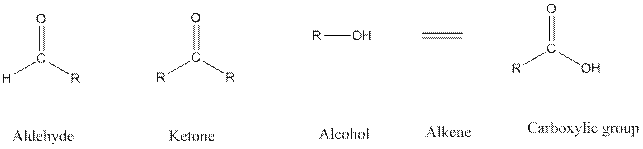
R represents any alkyl chain.
In functional group ketone carbonyl carbon is bonded to two alkyl groups, in aldehyde carbonyl carbon is attached to one alkyl group and one hydrogen atom, in alcohol functional group an −OH is bonded to an alkyl group.
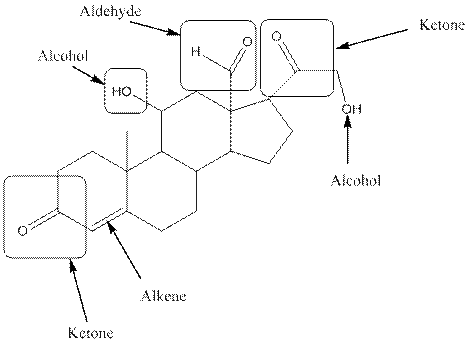
The compound aldosterone consists of two alcohol frunctional group, two ketone functional groups and one aldehyde functional group and one alkene. So, four types of functional group, that is, alcohol, aldehyde, ketone and alkene.
(b)
Interpretation:
All the lone pairs on oxygen atom of aldosterone should be drawn.
Concept Introduction:
Oxygen atom has six valence electrons. The electrons which are used in bond formation are called bonding electrons. The electrons that remain on an atom after bond formation are called lone pair electrons.
Answer to Problem 83P
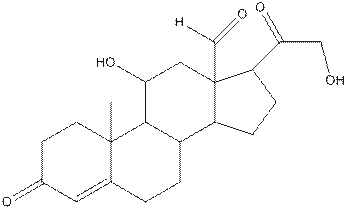
The structure of aldosterone having all lone pairs on oxygen atom is,
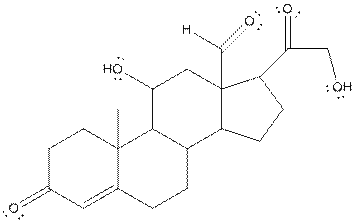
Explanation of Solution
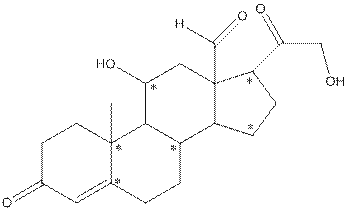
The structure of aldosterone is as follows:
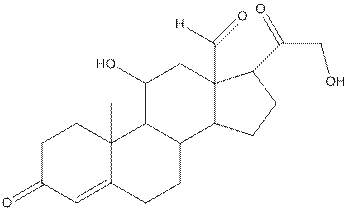
In the compound, three double bonded oxygen atoms present. Oxygen has six valence electrons. Two electrons in all double bonded oxygen atoms are used for formation of double bond. Remaining four electrons present as two lone pair. Also the compound contains three single bonded oxygen atoms. One electron is used for making O-H bond and one electron is used for making O-C bond. Remaining four electrons present as two lone pairs on each single bonded oxygen atom. Hence, the structure having all lone pairs on oxygen atom is,
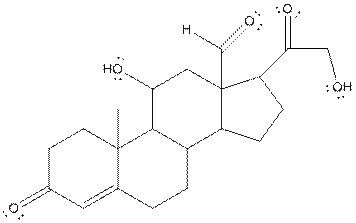
(c)
Interpretation:
The number of carbon atoms in the structure of aldosterone should be determined.
Concept Introduction:
In skeletal structure the terminals represent methyl
Answer to Problem 83P
Twenty one carbon atoms present in the aldosterone.
Explanation of Solution
The structure of aldosterone is given as follows:
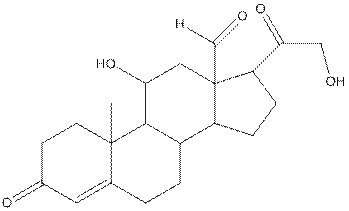
In skeletal structure the terminals represent methyl
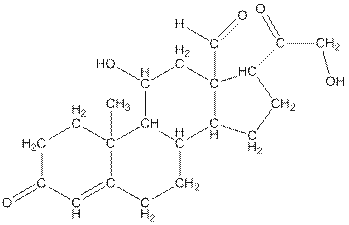
Hence, twenty one carbon atoms present in the compound.
(d)
Interpretation:
The number of hydrogen atoms present in the * carbon atoms in aldosterone should be determined.
Concept Introduction:
In skeletal structure the terminals represent methyl
Answer to Problem 83P
The * carbon atoms have total five hydrogen atoms.
Explanation of Solution
In skeletal structure the terminals represent methyl
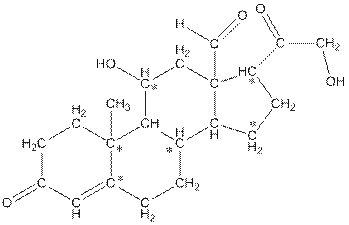
Hence, the * carbon atoms have total five hydrogen atoms.
(e)
Interpretation:
The shape around each indicated carbon atom in aldosterone should be determined.
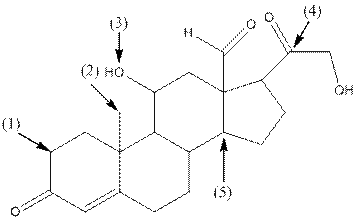
Concept Introduction:
The following table should be used while determining the shape around an atom.
| Number of groups | Number of atoms | Number of lone pairs | Shape | Bond angle |
| 2 | 2 | 0 | Linear | |
| 3 | 3 | 0 | Trigonal planar | |
| 4 | 4 | 0 | Tetrahedral | |
| 4 | 3 | 1 | Trigonal pyramidal | |
| 4 | 2 | 2 | Bent |
Answer to Problem 83P
- Shape around carbon (1) is tetrahedral, shape around carbon (2) is tetrahedral, shape around oxygen (3) is bent, shape around carbon (4) is trigonal planar and shape around carbon (5) is tetrahedral.
Explanation of Solution
The complete structure of aldosterone is as follows:
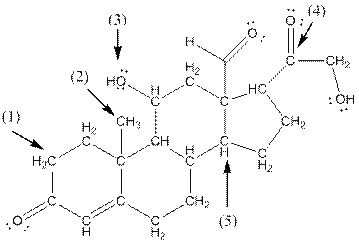
- Carbon (1) has four groups ( two hydrogen and two carbon) surrounding it. So, shape around carbon (1) is tetrahedral.
- Carbon (2) has four groups (three hydrogen and one carbon) surrounding it. So, shape around carbon (2) is tetrahedral.
- Oxygen (3) has two groups (one carbon and one hydrogen) and two lone pairs surrounding it. So, shape around oxygen (3) is bent.
- Carbon (4) has three groups (two carbon and one oxygen) surrounding it. So, shape around carbon (4) is trigonal planar.
- Carbon (5) has four groups (three carbon and one hydrogen) surrounding it. So, shape around carbon (5) is tetrahedral.
(f)
Interpretation:
All the polar bonds in aldosterone should be labeled.
Concept Introduction:
The unequal sharing of valence electrons in a bond is called polar bond. Polar bond result when the bond formed between two atoms in which one atom is more electronegative than the other one. One example of polar bond is
Structure of HCl is as follows:

In
Answer to Problem 83P
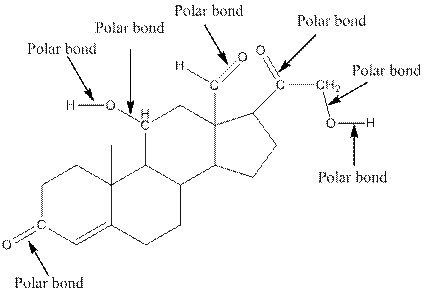
The structure of aldosterone with all polar bonds is as follows:
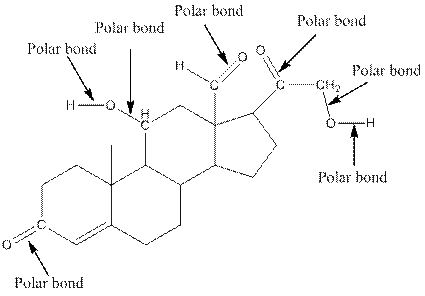
Explanation of Solution
In organic compound, most of the polar bonds formed between carbon and heteroatoms like oxygen, nitrogen, sulphur etc. To identify the polar bonds in the compound aldosterone, find the bonds between carbon and oxygen or oxygen and hydrogen because oxygen is more electronegative than carbon and oxygen is more electronegative than carbon. In aldostrone, five polar bonds are formed between carbon and oxygen (oxygen is more electronegative than carbon) and two polar bonds present between oxygen and hydrogen (oxygen is more electronegative than hydrogen). Thus, the polar bonds are:
Want to see more full solutions like this?
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardIdentify the functional groups labeled with an arrow. The only new name here is lactone: it means a cyclic ester. Also do not bother if it says 1° alcohol or 2° alcohol. Just look for the alcohol. OGMU 721514 Q I H3C 11 HO но. O O OI=1 alcohol; II = lactone; III = ether OI= ester; II = 2° alcohol; III = ether 1 = lactone; II = 2° alcohol; III = ester OI=2° alcohol; II = lactone; III = lactone 1 = ester; II = 2° alcohol; III = :lactone O O OH H HO OBnarrow_forwardCompare the two molecules shown. Choose the response that accurately describes how they are related to one another. СН3 сHз сHCHCH2C CH3 нз СН3 l CH₂ CH3CH2 C CH2CH3 | CH3 O The molecules are isomers O The molecules are the same O The molecules are not isomers (and are not the same molecule)arrow_forward
- uestion 6 The correct IUPAC name for this compound is It may help to draw the complete Lewis Structure. OH CH3CH(CH2),CH3 O 2-heptanol O 2-butanol O 2-hydroxybutane O 2-hydroxyl-2-butylbutane O heptanol A Moving to another question will save this response. MacBc 20 F2 F3 F4 F5 $4 2 4 Warrow_forwardFor the compounds below, which, if any, exist as cis- or trans-isomers? Type the number or numbers of the compound(s) (separated by a comma with no spaces). If none of the compounds exist as cis- or trans-isomers, type none. H₂C: 3 CH3 CH3 CH3 CH3 H₂C Cl 2 4 -CH3 CH3arrow_forwardThe structure of estrone, a female sex hormone, is shown below. Highlight and identify all of the functional groups. но Estrone Dihydroxyacetone (DHA), also known as glycerone, is the active ingredient in sunless tanning products. DHA has the same molecular formula as glyceraldehyde, an important intermediate in the metabolism of glucose. Highlight and label all the functional groups in DHA and glyceraldehyde. What is the molecular formula for DHA and glyceraldehyde? What type of isomers are DHA and glyceraldehyde? OH но, HO, LOH DHA Glyceraldehydearrow_forward
- List the name and position for each substituent in this organic molecule on a separate row of the table below: CH3CH2CH2Ọ CH3 CH3–CH2–CH–CH2–CH–CH2–CH2–CH3 Your answer for the name column should be just one word. In the position column, write the number of the carbon where the substituent is attached to the parent chain (which has been highlighted for you). Note for advanced students: You may assume that the parent chain is numbered from left-to-right in this molecule. You can add more rows to the table if you need them. name Add Row position 0 X Śarrow_forwardPart A 1 of 18 What is the name of the compound shown here? Enter the name of the compound. ► View Available Hint(s) > C3H7 C₂H₂ C₂H₂ OHarrow_forward0 hn. Ch3 oh. What is this formula molecular structure, number of carbon sites how many hydrogen sites how many atoms each How many atoms in each type of atom in the structure give a molecular formula and identify tw functional grarrow_forward
- Identify the functional group present in the following compounds. 1. CH;CH,CH,SH 2. CH,CH,CH,CH,NHCH; C ÇI 3. CH3 - CH -CH -CH,CH; CH3 CH ーで CH HC 4. CH CH CH 5. CH -С-0-С -СH-СНзarrow_forwardWrite the systematic name of each organic molecule: OH | structure CH3 -CH–CH2–CH2–CH3 HỌ—CH2−CH2 —CH,—OH HO—CH,—CH2−CH2 —CH3 name 0 0arrow_forwardDraw a skeletal ("line") structure of this molecule: HO—CH2 CH3 CH₂ CH₂-CH-CH3 Click and drag to start drawing a structure.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

