
Concept explainers
Convert each shorthand structure to a complete structure with all atoms and lone pairs drawn in.
-
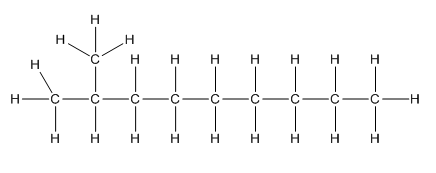
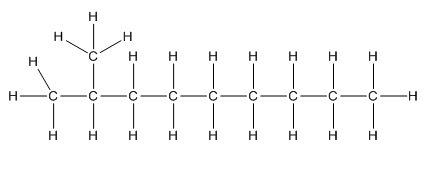
a.
b.
c.
d. 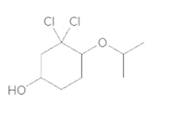
(a)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
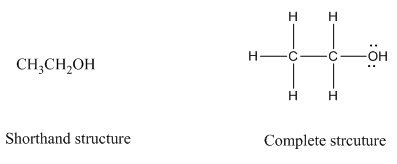
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
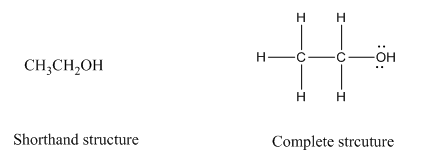
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
Complete structure of the compound is,
Explanation of Solution
Given compound is as follows:
To −CH group two methyl
(b)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
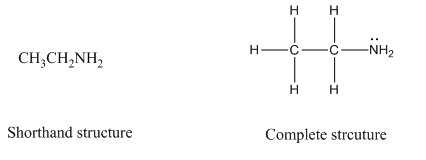
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
Complete structure of the compound is as follows:
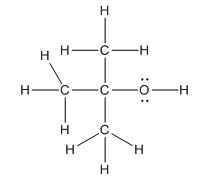
Explanation of Solution
In the compound
Hence, complete structure of the compound is as follows:
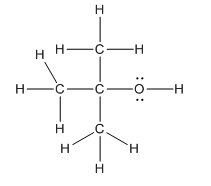
(c)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
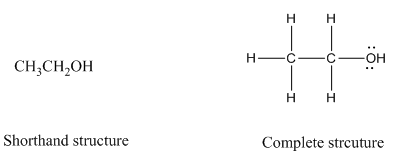
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
Complete structure of the compound is as follows:
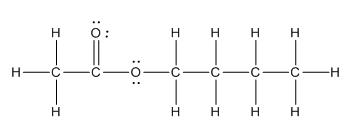
Explanation of Solution
Given compound is as follows:
In the complete structure, all the bonds between atoms and lone pairs are to be shown. Two oxygen atoms present. Both the oxygen atoms have two lone pairs (as the total number of valence electron of O is 6 and out of these 2 are involved in bond formation) on it. Hence, complete structure of the compound is as follows:
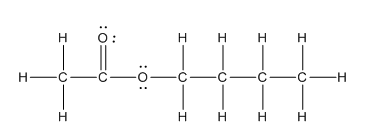
(d)
Interpretation:
To convert the following shorthand structure to complete structure with all atoms and lone pairs drawn.
Concept Introduction:
Complete structure of a compound is the one in which all the bonds, atoms and lone pairs are shown. Example of complete structure is given below.
In shorthand structure, bond between the atoms and lone pairs of atoms are not shown. But, in complete structure, all the bonds between atoms and lone pairs are shown.
Answer to Problem 37P
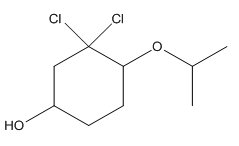
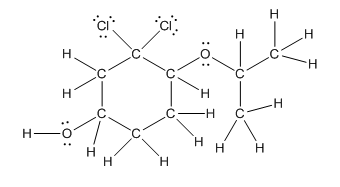
Complete structure is as follows:
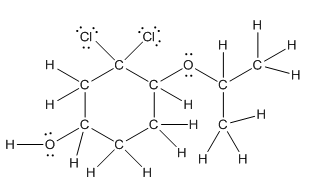
Explanation of Solution
Given compound is as follows:
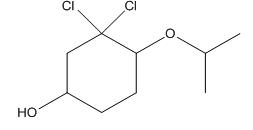
In complete structure, all the carbon atoms, hydrogen atoms and all the lone pairs should be shown. In the compound two chlorine atoms (as the total number of valence electron of Cl is 7 and out of these 1 is involved in bond formation), one oxygen atom and one −OH group(as the total number of valence electron of O is 6 and out of these 2 are involved in bond formation) present which has lone pairs. So, complete structure is as follows:
Want to see more full solutions like this?
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardFunctionalized Hydrocarbons Identify each compound according to its functional group (e.g.,amine,ester,etc.):arrow_forwardBased on the molecular formula, determine whether each compound is an alkane, alkene, or alkyne. (Assume that the hydro- carbons are noncyclical and there is no more than one multiple bond.) a. C5H12 b. C3H6 с. С-Н12 d. C11H22arrow_forward
- Which of the following compounds is a valid Lewis structure of a hydrocarbon? H H-C-Ö-H H HH H-C-N-H H I II H H-C-C-H H III HH H-C-C-H I НН IV I, II, and IV are valid structures of hydrocarbons. Only III is a valid structure of hydrocarbons. All of these compounds are valid structures of hydrocarbons. Only IV is a valid structure of hydrocarbons. III and IV are valid structures of hydrocarbons.arrow_forwardDraw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a diethyl ether, (CH3CH2)2O, the first general anesthetic used in medical procedures b. acrylonitrile, CH2CHCN, starting material used to manufacture synthetic Orlon fibers c.dihydroxyacetone, (HOCH2)2CO, an ingredient in sunless tanning products d.acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forwardIdentify the circled functional group(s) for each molecule. OH CH3CHCH3 a. ΙΟ ——CH2 b. NH2CH2CH2CH2NHCH3 C. H₂N d. 0 Circle and name all functional group(s) for each molecule CH3 HO OH SH CH2CHCH NHCH(CH3)2 OHarrow_forward
- Convert each molecule to a skeletal structure. a. (CH3)2CHCH2CH2CH(CH3)2 b. CH3CH(Cl)CH(OH)CH3 c.CH3(CH2)2C(CH3)2CH(CH3)CH(CH3)CH(Br)CH3arrow_forwardWhich of these formulas is(are) not valid for octane? A. C8H18B. C8H20C. C8H16D. CH3CH2CH2CH2CH2CH2CH2CH3E. CH3(CH2)6 CH3arrow_forwardDraw the cis and trans isomers of 2-butene, CH3CHCHCH₂. Show all hydrogen atoms. cis-2-butene Incorrect Y trans-2-butene Incorrect YA Harrow_forward
- CH2-CH2-CH3 Type of functional group r. CH2-CH2-CH3 Common Name Type of functional group H3C-C-o IUPAC H3C- Common Name H,C-CH,-CH,-CH,-CH,-NH-ċ-CH -CH3 t. Type of functional grouparrow_forwardConvert each condensed formula to a Lewis structure. CH3(CH2)4CH(CH3)2 (CH3)3CCH(OH)CH2CH3 (CH3)2CHCHO (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardWhat is the class of organic compound to which the molecule belong? A. H2N B. CH3 HC= CH3 C. CI, CI CI- CH3arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


